Philips Sonicare FlexCare - Sonicare.com - Sonicare
Philips Sonicare FlexCare - Sonicare.com - Sonicare
Philips Sonicare FlexCare - Sonicare.com - Sonicare
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
times as single cells and sometimes as larger<br />
colonies. The biofilm was approximately 50 µm<br />
thick. The laboratory-grown S mutans demonstrated<br />
similar characteristics of aggregated bacteria: interspersed<br />
with water channels and enveloped in a<br />
slime matrix.<br />
Biofilm Growth on Cellulose Ester Membranes<br />
S mutans biofilms grew readily on the cellulose<br />
ester membranes. The biofilms were similar to those<br />
described above and in the literature 16,18 and consisted<br />
of dense aggregates of cell clusters interspersed<br />
with water channels that penetrated to the membrane<br />
filter surface (Figure 4).<br />
The biofilms ranged in maximum thickness<br />
from 20 µm to 80 µm with an average of 45 µm ± 25<br />
µm (mean ± 1 SD) and had a surface coverage of<br />
between 50% and 75%.<br />
Delivery of Fluoride Through Biofilmcolonized<br />
Membranes With and Without<br />
Power Brushing<br />
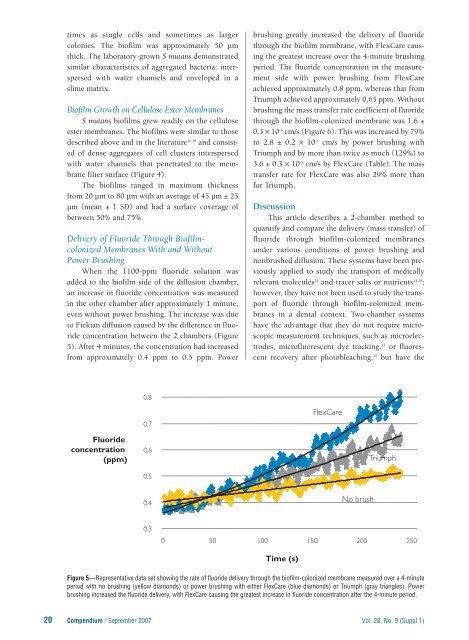
When the 1100-ppm fluoride solution was<br />
added to the biofilm side of the diffusion chamber,<br />
an increase in fluoride concentration was measured<br />
in the other chamber after approximately 1 minute,<br />
even without power brushing. The increase was due<br />
to Fickian diffusion caused by the difference in fluoride<br />
concentration between the 2 chambers (Figure<br />
5). After 4 minutes, the concentration had increased<br />
from approximately 0.4 ppm to 0.5 ppm. Power<br />
0.9<br />
brushing greatly increased the delivery of fluoride<br />
through the biofilm membrane, with <strong>FlexCare</strong> causing<br />
the greatest increase over the 4-minute brushing<br />
period. The fluoride concentration in the measurement<br />
side with power brushing from <strong>FlexCare</strong><br />
achieved approximately 0.8 ppm, whereas that from<br />
Triumph achieved approximately 0.65 ppm. Without<br />
brushing the mass transfer rate coefficient of fluoride<br />
through the biofilm-colonized membrane was 1.6 ±<br />
0.3 × 10 -5 cm/s (Figure 6). This was increased by 79%<br />
to 2.8 ± 0.2 × 10 -5 cm/s by power brushing with<br />
Triumph and by more than twice as much (129%) to<br />
3.6 ± 0.3 × 10 -5 cm/s by <strong>FlexCare</strong> (Table). The mass<br />
transfer rate for <strong>FlexCare</strong> was also 29% more than<br />
for Triumph.<br />
Discussion<br />
This article describes a 2-chamber method to<br />
quantify and <strong>com</strong>pare the delivery (mass transfer) of<br />
fluoride through biofilm-colonized membranes<br />
under various conditions of power brushing and<br />
nonbrushed diffusion. These systems have been previously<br />
applied to study the transport of medically<br />
relevant molecules 22 and tracer salts or nutrients 23,24 ;<br />
however, they have not been used to study the transport<br />
of fluoride through biofilm-colonized membranes<br />
in a dental context. Two-chamber systems<br />
have the advantage that they do not require microscopic<br />
measurement techniques, such as microelectrodes,<br />
microfluorescent dye tracking, 27 or fluorescent<br />
recovery after photobleaching, 22 but have the<br />
0.8<br />
0.7<br />
<strong>FlexCare</strong><br />
Fluoride<br />
concentration<br />
(ppm)<br />
0.6<br />
0.5<br />
Triumph<br />
0.4<br />
No brush<br />
0.3<br />
0 50 100 150 200 250<br />
Time (s)<br />
Figure 5—Representative data set showing the rate of fluoride delivery through the biofilm-colonized membrane measured over a 4-minute<br />
period with no brushing (yellow diamonds) or power brushing with either <strong>FlexCare</strong> (blue diamonds) or Triumph (gray triangles). Power<br />
brushing increased the fluoride delivery, with <strong>FlexCare</strong> causing the greatest increase in fluoride concentration after the 4-minute period.<br />
20 Compendium / September 2007 Vol. 28, No. 9 (Suppl 1)