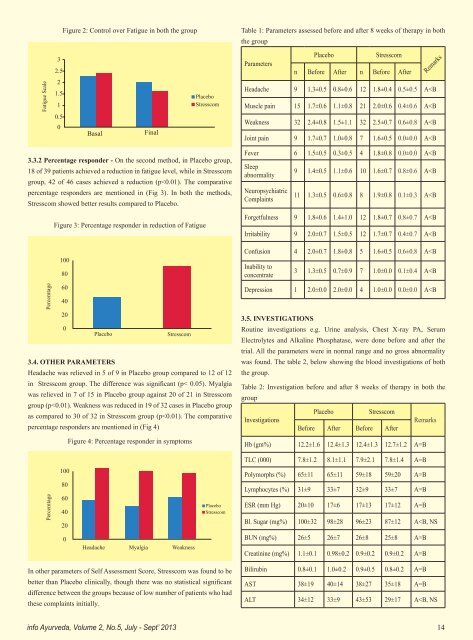
Figure 2: Control over Fatigue in both the group Table 1: Parameters assessed before and after 8 weeks of therapy in both the group Fatigue Scale 3 2.5 2 1.5 1 0.5 0 Basal Final Placebo Stresscom Placebo Stresscom Parameters n Before After n Before After Remarks Headache 9 1.3±0.5 0.8±0.6 12 1.8±0.4 0.5±0.5 A
3.6. TOLERANCE The drug was well <strong>to</strong>lerated in both the groups with no patient requiring withdrawal from therapy. In Placebo group, 3 patients had minor self limiting adverse effects in the form of nausea, vomiting and anorexia; 36 of 39 (92%) patients <strong>to</strong>lerated the placebo without any specfic safety concerns. In Stresscom group, 6 patients had side effects in the form of minor self limiting nausea in 3, heart burn in 1, belching with epigastric fullness in 1 and menorrhagia in 1 patient that was easily controlled with symp<strong>to</strong>matic therapy; 41 of 47 patients (87%) <strong>to</strong>lerated Stresscom without any specfic safety concerns. The two batches did not differ significantly in <strong>to</strong>lerance. 4. SUMMARY AND CONCLUSIONS The double blind randomized placebo controlled study was conducted in 86 patients of Chronic Fatigue Syndrome attending the Out Patient Department of LNJP Hospital, New Delhi, for a period of 8 <strong>to</strong> 12 weeks. The following points can be concluded basis the result of the clinical study: • Stresscom had shown improvement on multiple parameters of psychosomatic complaints of Chronic Fatigue Syndrome like fatigue, headache, muscle pain, weakness, joint pains, feverish feeling, sleep abnormality, neuropsychiatric complaints, f<strong>org</strong>etfulness, irritability, confusion, lack of concentration as compared <strong>to</strong> the baseline scores and that of placebo. A statistical significant improvement was noted in parameters of fatigue, headache, muscle pain and weakness compared <strong>to</strong> placebo. • Stresscom had shown improvement in the scores of Karnofsky Performance Scale Index (KPI) in each visit compared <strong>to</strong> the baseline, however, no significant difference was found on between the group analysis. • Both Stresscom and Placebo were found <strong>to</strong> be well <strong>to</strong>lerated during the course of study. • Stresscom might be useful in relieving symp<strong>to</strong>ms of Chronic Fatigue Syndrome. 5. ACKNOWLEDGEMENT Dabur India Limited is thankful <strong>to</strong> the Investiga<strong>to</strong>rs for conducting the study on the product Ashwagandha Capsule, which is being marketed in India by Dabur as Stresscom Capsules 6. REFERENCES • Medicinal Plants of India and Pakistan; Dastur, J.F, D.B . Taraporvala & Sons, Bombay; (1970), 3rd edition pp.177. • Role of Aswagandha in various types of Arthropathies; Bec<strong>to</strong>r, N.P., Puri, A.S. & Sharma, D: (1968) Ind. J. Med. Res. 56 (10), pp 1581- 1583. • Dimensional assessment of chronic fatigue syndrome; Jan H.M.M. Vercoulen, Caroline M.A. Swanink, Jan F.M. Fennis et.al;Journal of Psychosomatic Research; <strong>Volume</strong> 38, Issue 5, <strong>July</strong> 1994, Pages 383– 39 • Chronic fatigue syndrome (CFS) -A review and a proposition of a biopsycho-social model of the chronic fatigue syndrome; Rolf H. Adler; Swiss Med Wkly 2004;134:268–276 MAKE US STRONG, BECOME OUR MEMBER CATEGORY OF APPLICANT (Please the appropriate category) Affiliated Members Institutional Members Institutional Members Industries involved in manufacture and sale of herbal products or extracts. Affiliated Members : Regional or provincial Association of Manufacturers of Ayurvedic Medicines. Name of Association/ Firm/Institution: ________________________________________________________________________________________ Mailing Address:___________________________________________________________________________________________________________ Registered Address: _______________________________________________________________________________________________________ Phone No.: _____________________________________________________Mobile No. _______________________________________________ Fax: ___________________________________________ Email ID: _______________________________________________________________ Signature & date: _____________________________ Please enclose the Bank Draft in favour of “Association of Manufacturers of Ayurvedic Medicines” payable at New Delhi as per relevant category mentioned below and send the complete form <strong>to</strong> AMAM’s address. (Bank Draft No.________________Drawn on (Bank name)____________________________________ dated _________ for Rs._______________) FOR OFFICIAL USE ONLY Application Received on_______________ Members subscription Received_________________________________ Discussed in the meeting of Association. Dated ______________Membership No. Allotted_____________________________________________________________________ MEMBERSHIP FEE A. Institutional Members Turn over up<strong>to</strong> Rs. 10 Crores Rs. 2500/year Turn over Rs. 10 Crores and above Rs. 10000/year B. Affiliated membership for Organization/Federations Rs. 10000/year Pradeep Multani (General Secretary) Approved / Rejected may be enlarged and xeroxed Published by: Association of Manufacturers of Ayurvedic Medicines Regd. Office: 22 Site –IV, Sahibabad, Ghaziabad - 201010 (UP) Tel: 0120, 4378400, Fax: 0120 4376909 Correspondence Address: H-36, Connaught Place, New Delhi-110001 Tel: 011-23350062, Fax: 011-23350063 e-mail : <strong>amam</strong>india@gmail.com website: www.<strong>amam</strong>-<strong>ayurveda</strong>.<strong>org</strong>












![[To be published in Gazette of India Part II Section 3, sub-section iii]](https://img.yumpu.com/28570283/1/190x245/to-be-published-in-gazette-of-india-part-ii-section-3-sub-section-iii.jpg?quality=85)


