Atomic structure, bonding
Atomic structure, bonding
Atomic structure, bonding
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
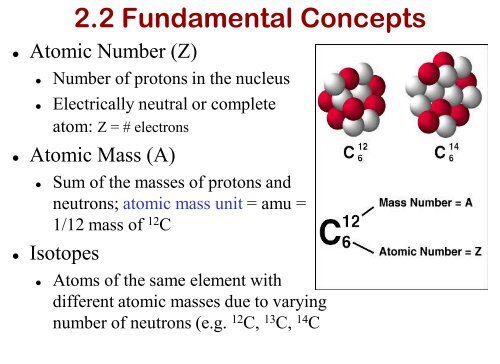
2.2 Fundamental Concepts<br />
• <strong>Atomic</strong> Number (Z)<br />
• Number of protons in the nucleus<br />
• Electrically neutral or complete<br />
atom: Z = # electrons<br />
• <strong>Atomic</strong> Mass (A)<br />
• Sum of the masses of protons and<br />
neutrons; atomic mass unit = amu =<br />
1/12 mass of 12 C<br />
• Isotopes<br />
• Atoms of the same element with<br />
different atomic masses due to varying<br />
number of neutrons (e.g. 12 C, 13 C, 14 C