Lumax Family of ICDs and CRTâDs - BIOTRONIK USA - News
Lumax Family of ICDs and CRTâDs - BIOTRONIK USA - News
Lumax Family of ICDs and CRTâDs - BIOTRONIK USA - News
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
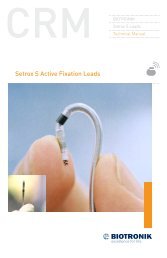
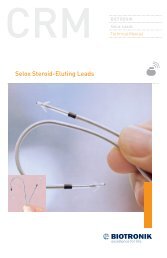
2 <strong>Lumax</strong> <strong>Family</strong> Technical Manual<br />
• <strong>Lumax</strong> VR—provides single chamber rate adaptive bradycardia<br />
pacing support as well as tachyarrhythmia detection <strong>and</strong> therapy.<br />
• <strong>Lumax</strong> VR‐T—In addition to the functionality found with st<strong>and</strong>ard<br />
VR model, it also has <strong>BIOTRONIK</strong>’s Home Monitoring System.<br />
The Home Monitoring System enables automatic exchange <strong>of</strong><br />
information about a patient’s cardiac status from the implant to the<br />
physician remotely.<br />
• <strong>Lumax</strong> VR-T DX – provides ventricular rate adaptive bradycardia<br />
pacing support that can include atrial tracking with a single pass<br />
ICD lead <strong>and</strong> also has <strong>BIOTRONIK</strong>’s Home Monitoring system.<br />
The 300⁄500 <strong>and</strong> 340⁄540 designation for each <strong>of</strong> the above‐described<br />
models denote the maximum programmable shock energy <strong>of</strong> 30 joules<br />
<strong>and</strong> 40 joules, respectively.<br />
The <strong>Lumax</strong> 500⁄540 models also feature a third programmable shock<br />
path for delivery <strong>of</strong> defibrillation⁄cardioversion shocks. The shock path<br />
is programmable between the different shock coils (SVC⁄RV) <strong>and</strong>⁄or the<br />
device housing. Section 2.7.3.6 provides further details on the available<br />
shock configurations.<br />
Additionally, the <strong>Lumax</strong> 500⁄540 models feature Automatic Threshold<br />
Measurement (ATM) <strong>of</strong> ventricular pacing thresholds. This feature<br />
is separately programmable for the right (RV) <strong>and</strong> left (LV) ventricle.<br />
Section 2.3 provides further details.<br />
The <strong>Lumax</strong> HF (‐T) models have three IS‐1 pacing⁄sensing header ports<br />
<strong>and</strong> two DF‐1 defibrillation⁄cardioversion ports. The <strong>Lumax</strong> DR (‐T)<br />
models have two IS‐1 pacing⁄sensing header ports. The <strong>Lumax</strong> VR (‐T)<br />
models have one IS‐1 pacing⁄sensing header ports. IS‐1 refers to the<br />
international st<strong>and</strong>ard whereby leads <strong>and</strong> generators from different<br />
manufacturers are assured a basic fit [Reference ISO 5841‐3:1992].<br />
DF‐1 refers to the international st<strong>and</strong>ard for defibrillation lead<br />
connectors [Reference ISO 11318:1993].<br />
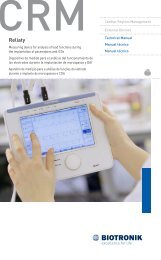
External devices that interact with <strong>and</strong> test the implantable devices are<br />
also part <strong>of</strong> the ICD⁄CRT‐D System. These external devices include the<br />
ICS 3000 Programming <strong>and</strong> Tachyarrhythmia Monitoring System <strong>and</strong><br />
the Implant Module System Analyzer or Pacing System Analyzer for<br />
acute lead testing. The ICS 3000 programmer is used to interrogate <strong>and</strong><br />
program the ICD⁄CRT‐Ds.<br />
<strong>BIOTRONIK</strong> conducted the TRUST study to evaluate the safety <strong>and</strong><br />
effectiveness <strong>of</strong> Home Monitoring, which is available in some models <strong>of</strong><br />
this device. Refer to Section 1.6.4 for details regarding the study design