Safety, Quality, Efficacy: Regulating Medicines in the UK
Safety, Quality, Efficacy: Regulating Medicines in the UK
Safety, Quality, Efficacy: Regulating Medicines in the UK
- No tags were found...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
SAFETY, QUALITY, EFFICACY: REGULATING MEDICINES IN THE <strong>UK</strong><br />
4.11 Industry representatives we spoke to felt <strong>the</strong>re was scope<br />
for <strong>the</strong> Agency to do more to promote <strong>the</strong> <strong>UK</strong> position<br />
<strong>in</strong> Europe and that more resources should be devoted to<br />
provid<strong>in</strong>g high-level representation on European expert<br />
committees. They would like to see European agencies<br />
generally tak<strong>in</strong>g a more <strong>in</strong>tegrated approach to<br />
regulation, with different agencies concentrat<strong>in</strong>g on<br />
<strong>the</strong>ir particular strengths, and rely<strong>in</strong>g more on each<br />
o<strong>the</strong>r's work.<br />
The Agency may be at risk of not<br />
provid<strong>in</strong>g services that <strong>in</strong>dustry<br />
with<strong>in</strong> Europe requires<br />
4.12 With an <strong>in</strong>creas<strong>in</strong>gly global pharmaceutical market,<br />
most new medic<strong>in</strong>es will be sold <strong>in</strong> more than one<br />
European country. It <strong>the</strong>refore makes sense for<br />
companies ei<strong>the</strong>r to apply to <strong>the</strong> European <strong>Medic<strong>in</strong>es</strong><br />
Evaluation Agency for a centralised European market<strong>in</strong>g<br />
authorisation, or to achieve mutual recognition of a<br />
licence obta<strong>in</strong>ed <strong>in</strong> one member state. Historically, <strong>the</strong><br />
<strong>UK</strong> has been among a handful of countries with wellestablished<br />
and well-respected regulatory expertise. The<br />
<strong>Medic<strong>in</strong>es</strong> Control Agency has as a result received a<br />
larger share of applications under <strong>the</strong> mutual<br />
recognition arrangements than would be expected.<br />
for <strong>in</strong>spection and enforcement activities. The Agency is<br />
acutely aware that it needs to rema<strong>in</strong> competitive to<br />
reta<strong>in</strong> its share of both centralised and mutual<br />
recognition bus<strong>in</strong>ess <strong>in</strong> Europe. However, <strong>the</strong> level and<br />
structure of fees charged by <strong>the</strong> Agency is a policy<br />
decision for M<strong>in</strong>isters.<br />
The Agency has worked with<br />
potentially conflict<strong>in</strong>g dual<br />
objectives to help <strong>in</strong>dustry and<br />
protect public health<br />
4.15 S<strong>in</strong>ce its creation, <strong>the</strong> Agency has been required <strong>in</strong> its<br />
Framework Document to avoid creat<strong>in</strong>g "unnecessary<br />
impediments" to <strong>the</strong> pharmaceutical <strong>in</strong>dustry. In l<strong>in</strong>e<br />
with this, <strong>the</strong> Agency has recently been closely <strong>in</strong>volved<br />
<strong>in</strong> efforts to improve <strong>the</strong> regulatory environment for<br />
<strong>in</strong>dustry through <strong>the</strong> work of <strong>the</strong> Pharmaceutical<br />
Industry Competitiveness Task Force, set up by <strong>the</strong><br />
Government <strong>in</strong> March 2000. This role has, however,<br />
been articulated <strong>in</strong> <strong>the</strong> Agency's 2001 Corporate Plan <strong>in</strong><br />
a more active way than its orig<strong>in</strong>al remit not to create<br />
impediments. The remit has been re-expressed as be<strong>in</strong>g<br />
"to facilitate <strong>the</strong> development of a successful <strong>UK</strong><br />
pharmaceutical <strong>in</strong>dustry for <strong>the</strong> benefit of <strong>the</strong> wider<br />
<strong>in</strong>terest of <strong>the</strong> <strong>UK</strong> economy".<br />
4.13 In recent years, however, competition from o<strong>the</strong>r<br />
European regulators has <strong>in</strong>creased. The two ma<strong>in</strong><br />
reasons for this, put to us by <strong>in</strong>dustry <strong>in</strong> <strong>the</strong> <strong>UK</strong> and<br />
regulators abroad, were:<br />
! that <strong>in</strong>dustry is <strong>in</strong>creas<strong>in</strong>gly look<strong>in</strong>g to consult with<br />
regulatory agencies for advice before submitt<strong>in</strong>g<br />
applications. While <strong>the</strong> Swedish Medical Products<br />
Agency has focused on encourag<strong>in</strong>g dialogue with<br />
companies, and charges for its advice, <strong>the</strong><br />
<strong>Medic<strong>in</strong>es</strong> Control Agency has not been able to<br />
charge a fee for advice to <strong>in</strong>dustry. As a result, it has<br />
found it difficult to devote resources to this work.<br />
The Agency is consider<strong>in</strong>g what changes to its<br />
powers may be needed to allow it to charge fees for<br />
this work; and<br />
! <strong>the</strong> fees charged by <strong>the</strong> <strong>Medic<strong>in</strong>es</strong> Control Agency<br />
for new active substances are <strong>the</strong> highest national<br />
fees <strong>in</strong> Europe.<br />
4.14 The high level of <strong>UK</strong> licence fees partly reflects <strong>the</strong> fact<br />
that <strong>the</strong> <strong>Medic<strong>in</strong>es</strong> Control Agency is f<strong>in</strong>anced entirely<br />
by fee <strong>in</strong>come, whilst some o<strong>the</strong>r regulators have partial<br />
state fund<strong>in</strong>g. It also reflects <strong>the</strong> fact that <strong>UK</strong> headl<strong>in</strong>e<br />
fees are designed to cover a broader range of work<br />
throughout <strong>the</strong> life of a product than is <strong>the</strong> case <strong>in</strong> some<br />
o<strong>the</strong>r countries. For example, <strong>the</strong> Ne<strong>the</strong>rlands<br />
<strong>Medic<strong>in</strong>es</strong> Evaluation Board Agency is not responsible<br />
4.16 Stakeholders from <strong>the</strong> consumer and patient groups we<br />
consulted were concerned that <strong>the</strong> relationship between<br />
<strong>the</strong> Agency and <strong>the</strong> <strong>in</strong>dustry it regulates may be too<br />
close. This perception is re<strong>in</strong>forced by <strong>the</strong> fact that <strong>the</strong><br />
Agency is fully funded by fees from <strong>in</strong>dustry, which is<br />
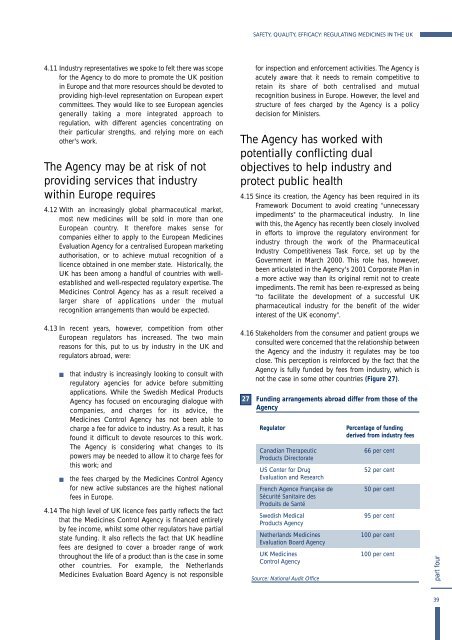
not <strong>the</strong> case <strong>in</strong> some o<strong>the</strong>r countries (Figure 27).<br />
27<br />
Fund<strong>in</strong>g arrangements abroad differ from those of <strong>the</strong><br />
Agency<br />
Regulator<br />
Canadian Therapeutic<br />
Products Directorate<br />
US Center for Drug<br />
Evaluation and Research<br />
French Agence Française de<br />
Sécurité Sanitaire des<br />
Produits de Santé<br />
Swedish Medical<br />
Products Agency<br />
Ne<strong>the</strong>rlands <strong>Medic<strong>in</strong>es</strong><br />
Evaluation Board Agency<br />
<strong>UK</strong> <strong>Medic<strong>in</strong>es</strong><br />
Control Agency<br />
Source: National Audit Office<br />
Percentage of fund<strong>in</strong>g<br />
derived from <strong>in</strong>dustry fees<br />
66 per cent<br />
52 per cent<br />
50 per cent<br />
95 per cent<br />
100 per cent<br />
100 per cent<br />
part four<br />
39