Bulletin of the Sea Fisheries Institute 2 (156) 2002 - CEEMaR
Bulletin of the Sea Fisheries Institute 2 (156) 2002 - CEEMaR
Bulletin of the Sea Fisheries Institute 2 (156) 2002 - CEEMaR
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
52 K. FORMICKI, A. KORZELECKA-ORKISZ, M. BONIS£AWSKA, A. TAÑSKI, W. WAWRZYNIAK and A. WINNICKI<br />
number <strong>of</strong> movements [1min.]<br />
60<br />
50<br />
40<br />
30<br />
20<br />
10<br />
heart<br />
somatic<br />
0<br />
57 63 70 83 96 121 145 171<br />
time from fertilization [h]<br />
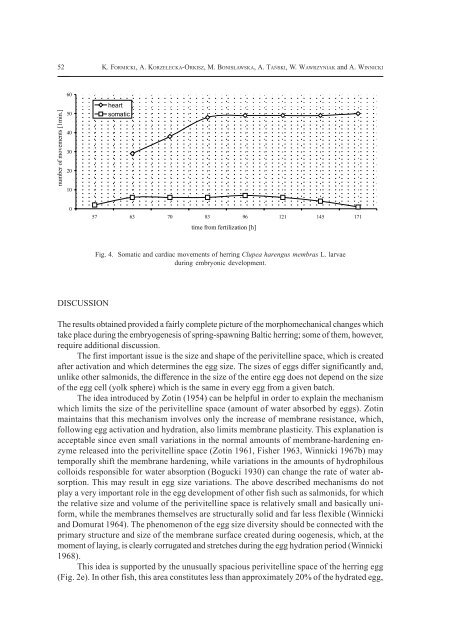
Fig. 4. Somatic and cardiac movements <strong>of</strong> herring Clupea harengus membras L. larvae<br />
during embryonic development.<br />
DISCUSSION<br />
The results obtained provided a fairly complete picture <strong>of</strong> <strong>the</strong> morphomechanical changes which<br />
take place during <strong>the</strong> embryogenesis <strong>of</strong> spring-spawning Baltic herring; some <strong>of</strong> <strong>the</strong>m, however,<br />
require additional discussion.<br />
The first important issue is <strong>the</strong> size and shape <strong>of</strong> <strong>the</strong> perivitelline space, which is created<br />
after activation and which determines <strong>the</strong> egg size. The sizes <strong>of</strong> eggs differ significantly and,<br />
unlike o<strong>the</strong>r salmonids, <strong>the</strong> difference in <strong>the</strong> size <strong>of</strong> <strong>the</strong> entire egg does not depend on <strong>the</strong> size<br />
<strong>of</strong> <strong>the</strong> egg cell (yolk sphere) which is <strong>the</strong> same in every egg from a given batch.<br />
The idea introduced by Zotin (1954) can be helpful in order to explain <strong>the</strong> mechanism<br />
which limits <strong>the</strong> size <strong>of</strong> <strong>the</strong> perivitelline space (amount <strong>of</strong> water absorbed by eggs). Zotin<br />
maintains that this mechanism involves only <strong>the</strong> increase <strong>of</strong> membrane resistance, which,<br />
following egg activation and hydration, also limits membrane plasticity. This explanation is<br />
acceptable since even small variations in <strong>the</strong> normal amounts <strong>of</strong> membrane-hardening enzyme<br />
released into <strong>the</strong> perivitelline space (Zotin 1961, Fisher 1963, Winnicki 1967b) may<br />
temporally shift <strong>the</strong> membrane hardening, while variations in <strong>the</strong> amounts <strong>of</strong> hydrophilous<br />
colloids responsible for water absorption (Bogucki 1930) can change <strong>the</strong> rate <strong>of</strong> water absorption.<br />
This may result in egg size variations. The above described mechanisms do not<br />
play a very important role in <strong>the</strong> egg development <strong>of</strong> o<strong>the</strong>r fish such as salmonids, for which<br />
<strong>the</strong> relative size and volume <strong>of</strong> <strong>the</strong> perivitelline space is relatively small and basically uniform,<br />
while <strong>the</strong> membranes <strong>the</strong>mselves are structurally solid and far less flexible (Winnicki<br />
and Domurat 1964). The phenomenon <strong>of</strong> <strong>the</strong> egg size diversity should be connected with <strong>the</strong><br />
primary structure and size <strong>of</strong> <strong>the</strong> membrane surface created during oogenesis, which, at <strong>the</strong><br />
moment <strong>of</strong> laying, is clearly corrugated and stretches during <strong>the</strong> egg hydration period (Winnicki<br />
1968).<br />
This idea is supported by <strong>the</strong> unusually spacious perivitelline space <strong>of</strong> <strong>the</strong> herring egg<br />
(Fig. 2e). In o<strong>the</strong>r fish, this area constitutes less than approximately 20% <strong>of</strong> <strong>the</strong> hydrated egg,