Scientific Article | Special IssueSp<strong>in</strong>al Cord Stimulation as a Method of Reduc<strong>in</strong>gOpioids <strong>in</strong> Severe Chronic Pa<strong>in</strong>: A Case Report andReview of the LiteratureTimothy Deer, MDPresident and CEO, The Center for Pa<strong>in</strong> Relief,CharlestonChristopher Kim, MDVice President, The Center for Pa<strong>in</strong> Relief, CharlestonRichard Bowman, MDPartner, The Center for Pa<strong>in</strong> Relief, CharlestonMatthew Ranson, MDPhysician, The Center for Pa<strong>in</strong> Relief, CharlestonC. Douglas Stewart PA-CDirector of Interventional Therapies, The Center forPa<strong>in</strong> ReliefWilfrido Tolent<strong>in</strong>o, PA-CDirector of Cl<strong>in</strong>ical Monitor<strong>in</strong>g, The Center for Pa<strong>in</strong>Relief, CharlestonAbstractOpioid addiction and abuse aregrow<strong>in</strong>g problems <strong>in</strong> the United <strong>State</strong>s,particularly <strong>in</strong> Appalachian areas, 13 whichhas led to a major social health problemcost<strong>in</strong>g millions of dollars <strong>in</strong> lost wages,medical care and lost productivity. In somepatients with chronic moderate to severepa<strong>in</strong>, opioids are <strong>in</strong>dicated and can besuccessfully used with proper monitor<strong>in</strong>g.In this report, we present a case where theuse of sp<strong>in</strong>al cord stimulation (SCS) led toan elim<strong>in</strong>ation of opioids, a return to work,and to productive function. We also reviewthe literature on the use of SCS to reduceopioid use and improve function based onobjective criteria.IntroductionSp<strong>in</strong>al Cord Stimulation (SCS)was first described <strong>in</strong> the literature<strong>in</strong> 1967, when it was used by Shealyto treat pa<strong>in</strong> secondary to <strong>in</strong>vasivecancer <strong>in</strong>volv<strong>in</strong>g the thoracicnerve roots. 1 Shortly after this<strong>in</strong>itial report, the Food and DrugAdm<strong>in</strong>istration (FDA) approvedthe use of SCS for neuropathic pa<strong>in</strong>of the trunk and limbs. Prospectivestudies have found the successfuluse of this modality <strong>in</strong> patientssuffer<strong>in</strong>g from failed back surgerysyndrome, diabetic neuropathy,complex regional pa<strong>in</strong> syndrome,cervical and lumbar radiculopathy,post herpetic neuralgia, trigem<strong>in</strong>alneuralgia, ischemic limb pa<strong>in</strong>, and<strong>in</strong>tractable ang<strong>in</strong>a. 2,3,4 This therapyhas been shown to be most effective<strong>in</strong> patients who describe their pa<strong>in</strong>as burn<strong>in</strong>g, stabb<strong>in</strong>g, shoot<strong>in</strong>g,and/or throbb<strong>in</strong>g. Once the deviceis implanted the patients are placed<strong>in</strong> a comprehensive rehabilitationprogram to improve function.Case ReportThe patient was a 28 year oldmale who was <strong>in</strong>jured <strong>in</strong> the courseof a skydiv<strong>in</strong>g misadventure. Hesuffered severe trauma to his aorta,visceral organs, and sp<strong>in</strong>e. His sp<strong>in</strong>altrauma led to extensive surgicalreconstruction of his lumbar sp<strong>in</strong>eand sacrum, which <strong>in</strong> turn led tosevere pa<strong>in</strong> from failed back surgerysyndrome and arachnoiditis. Hisfunction was very limited and hewas unable to walk <strong>in</strong>dependently,requir<strong>in</strong>g a wheelchair. Hismedications <strong>in</strong>cluded Methadone200 mgs per day, Gabapent<strong>in</strong> 4 gramsper day, and baclofen 80mgs per day.The methadone dose is consideredvery high for chronic non-cancerrelated pa<strong>in</strong> syndromes. His sideeffects from his oral pa<strong>in</strong> regimen<strong>in</strong>cluded sedation, constipation,irritability, and fatigue. At thispo<strong>in</strong>t the patient was referred forconsideration of a sp<strong>in</strong>al cordstimulation system. He met allcriteria set forth <strong>in</strong> the FDA approvaland was found to be psychologicallystable. Psychological clearance<strong>in</strong>cluded the absence of severeuntreated depression and anxiety. Healso did not demonstrate any f<strong>in</strong>d<strong>in</strong>gssuggestive of personality disorders.A sp<strong>in</strong>al cord stimulation systemis placed <strong>in</strong> two steps. Initially, a trialis performed by plac<strong>in</strong>g temporaryleads <strong>in</strong>to the epidural space toevaluate the patient’s response toelectrical current to the sp<strong>in</strong>al cord.The leads are placed <strong>in</strong> an attemptto create a t<strong>in</strong>gl<strong>in</strong>g sensation orparesthesia <strong>in</strong> the area of neuropathicpa<strong>in</strong>. The paresthesia is created bya balance of strategically arrangedcathodes and anodes that activatesp<strong>in</strong>al fibers <strong>in</strong>volved <strong>in</strong> the pa<strong>in</strong>pathways. The patient was educatedabout the goals of both the subjectiveoutcomes of pa<strong>in</strong> relief of 50% ormore, and the mechanical goals ofObjectivesThe objective of this submission is to provide <strong>in</strong>formation regard<strong>in</strong>g the efficacy of Sp<strong>in</strong>al Cord Stimulation, and the positive effectsof this therapy, <strong>in</strong>clud<strong>in</strong>g reduced dependence on opioids for the management of chronic pa<strong>in</strong> conditions.56 <strong>West</strong> Virg<strong>in</strong>ia <strong>Medical</strong> Journal
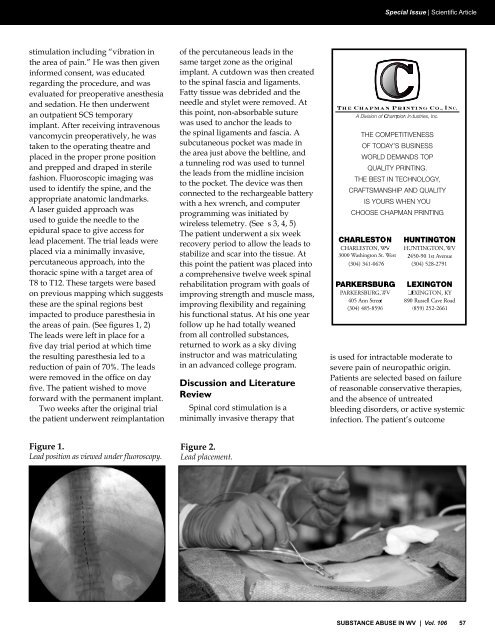
Special Issue | Scientific Articlestimulation <strong>in</strong>clud<strong>in</strong>g “vibration <strong>in</strong>the area of pa<strong>in</strong>.” He was then given<strong>in</strong>formed consent, was educatedregard<strong>in</strong>g the procedure, and wasevaluated for preoperative anesthesiaand sedation. He then underwentan outpatient SCS temporaryimplant. After receiv<strong>in</strong>g <strong>in</strong>travenousvancomyc<strong>in</strong> preoperatively, he wastaken to the operat<strong>in</strong>g theatre andplaced <strong>in</strong> the proper prone positionand prepped and draped <strong>in</strong> sterilefashion. Fluoroscopic imag<strong>in</strong>g wasused to identify the sp<strong>in</strong>e, and theappropriate anatomic landmarks.A laser guided approach wasused to guide the needle to theepidural space to give access forlead placement. The trial leads wereplaced via a m<strong>in</strong>imally <strong>in</strong>vasive,percutaneous approach, <strong>in</strong>to thethoracic sp<strong>in</strong>e with a target area ofT8 to T12. These targets were basedon previous mapp<strong>in</strong>g which suggeststhese are the sp<strong>in</strong>al regions bestimpacted to produce paresthesia <strong>in</strong>the areas of pa<strong>in</strong>. (See figures 1, 2)The leads were left <strong>in</strong> place for afive day trial period at which timethe result<strong>in</strong>g paresthesia led to areduction of pa<strong>in</strong> of 70%. The leadswere removed <strong>in</strong> the office on dayfive. The patient wished to moveforward with the permanent implant.Two weeks after the orig<strong>in</strong>al trialthe patient underwent reimplantationof the percutaneous leads <strong>in</strong> thesame target zone as the orig<strong>in</strong>alimplant. A cutdown was then createdto the sp<strong>in</strong>al fascia and ligaments.Fatty tissue was debrided and theneedle and stylet were removed. Atthis po<strong>in</strong>t, non-absorbable suturewas used to anchor the leads tothe sp<strong>in</strong>al ligaments and fascia. Asubcutaneous pocket was made <strong>in</strong>the area just above the beltl<strong>in</strong>e, anda tunnel<strong>in</strong>g rod was used to tunnelthe leads from the midl<strong>in</strong>e <strong>in</strong>cisionto the pocket. The device was thenconnected to the rechargeable batterywith a hex wrench, and computerprogramm<strong>in</strong>g was <strong>in</strong>itiated bywireless telemetry. (See s 3, 4, 5)The patient underwent a six weekrecovery period to allow the leads tostabilize and scar <strong>in</strong>to the tissue. Atthis po<strong>in</strong>t the patient was placed <strong>in</strong>toa comprehensive twelve week sp<strong>in</strong>alrehabilitation program with goals ofimprov<strong>in</strong>g strength and muscle mass,improv<strong>in</strong>g flexibility and rega<strong>in</strong><strong>in</strong>ghis functional status. At his one yearfollow up he had totally weanedfrom all controlled substances,returned to work as a sky div<strong>in</strong>g<strong>in</strong>structor and was matriculat<strong>in</strong>g<strong>in</strong> an advanced college program.Discussion and LiteratureReviewSp<strong>in</strong>al cord stimulation is am<strong>in</strong>imally <strong>in</strong>vasive therapy that3000 Wash<strong>in</strong>gton St. <strong>West</strong>is used for <strong>in</strong>tractable moderate tosevere pa<strong>in</strong> of neuropathic orig<strong>in</strong>.Patients are selected based on failureof reasonable conservative therapies,and the absence of untreatedbleed<strong>in</strong>g disorders, or active systemic<strong>in</strong>fection. The patient’s outcomeFigure 1.Lead position as viewed under fluoroscopy.Figure 2.Lead placement.SUBSTANCE ABUSE IN <strong>WV</strong> | Vol. 106 57