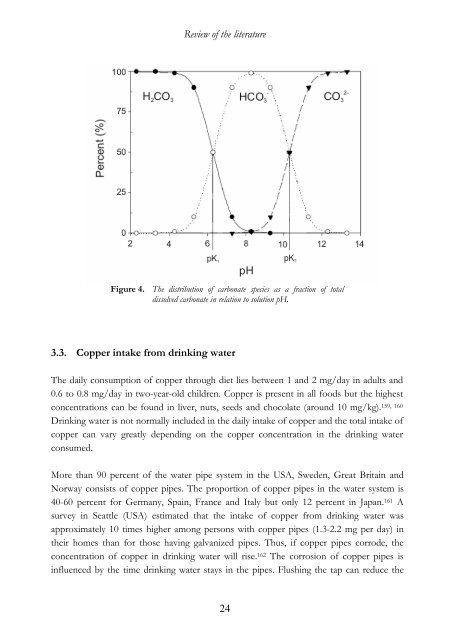
Review <strong>of</strong> the literature3.2. The role <strong>of</strong> bicarbonate and pH <strong>in</strong> dr<strong>in</strong>k<strong>in</strong>g <strong>water</strong>To prepare safe dr<strong>in</strong>k<strong>in</strong>g <strong>water</strong>, the <strong>water</strong> plants have to analyse which ions and chemicalcompounds are present <strong>in</strong> the <strong>water</strong>. The most important parameters for dr<strong>in</strong>k<strong>in</strong>g <strong>water</strong> arehardness, pH (alkal<strong>in</strong>ity and acidity) and sal<strong>in</strong>ity. The presence and comb<strong>in</strong>ation <strong>of</strong> anumber <strong>of</strong> ions will determ<strong>in</strong>e these features. Calcium and magnesium ions give thehardness, while bicarbonate, carbonate and hydroxyl ions give the alkal<strong>in</strong>ity <strong>in</strong> <strong>water</strong>.Chloride and sulphate ions (as HCl and H 2SO 4) contribute to the acidity and sal<strong>in</strong>ity <strong>of</strong> the<strong>water</strong> (Table 3, guidel<strong>in</strong>e values). Thus, calcium carbonate (CaCO 3) contributes both tohardness and alkal<strong>in</strong>ity <strong>of</strong> <strong>water</strong>, while calcium chloride (CaCl 2) gives hardness and sal<strong>in</strong>ity.The pH <strong>of</strong> dr<strong>in</strong>k<strong>in</strong>g <strong>water</strong> usually orig<strong>in</strong>ates from dissolved carbon dioxide (CO 2), whichforms carbonic acid (H 2CO 3). Humic acids and other organic acids, which orig<strong>in</strong>ate fromdecayed plants, can also make the <strong>water</strong> acidic. Acidification <strong>of</strong> the ground <strong>water</strong> <strong>in</strong>comb<strong>in</strong>ation with a bedrock that cannot counteract or buffer acidic particles will lead toacidic <strong>water</strong>. When the acidity orig<strong>in</strong>ates from natural sources, the pH <strong>of</strong> the <strong>water</strong> isaround 3-4. It is not harmful to dr<strong>in</strong>k such mildly acidic (pH~ 3-4) dr<strong>in</strong>k<strong>in</strong>g <strong>water</strong>.However, <strong>in</strong>directly, such acidic <strong>water</strong> will cause problems as it can extract metals from thesoil and <strong>water</strong> pipe systems. Acidic <strong>water</strong> can extract iron, manganese, copper, alum<strong>in</strong>iumand heavy metals such as cadmium and lead. Therefore, <strong>water</strong> plants adjust the pH to 6.5-9.5 (European guidel<strong>in</strong>e values, Council Directive 98/83/EC). Contribut<strong>in</strong>g factors <strong>in</strong><strong>in</strong>creased copper corrosion from copper pipes are the concentration <strong>of</strong> sulphate andchloride ions. 156Acidic <strong>water</strong> is usually adjusted to more alkal<strong>in</strong>e pH values with calcium carbonate(CaCO 3). 156-158 Alkal<strong>in</strong>ity is a measure <strong>of</strong> susceptibility for acidification, <strong>in</strong> other wordsbuffer capacity for acid addition. The higher the alkal<strong>in</strong>ity <strong>in</strong> the <strong>water</strong>, the better the <strong>water</strong>can resist acidification. Bicarbonate, carbonate and hydroxyl ions give different alkal<strong>in</strong>ity tothe <strong>water</strong>. If the pH <strong>in</strong> dr<strong>in</strong>k<strong>in</strong>g <strong>water</strong> is below 8.3, bicarbonate alkal<strong>in</strong>ity is prevail<strong>in</strong>g. Inmost dr<strong>in</strong>k<strong>in</strong>g <strong>water</strong> that has a pH between pH 5 and 8, the alkal<strong>in</strong>ity and pH come frombicarbonate ions (Figure 4). Bicarbonate and carbonate alkal<strong>in</strong>ity will exist <strong>in</strong> dr<strong>in</strong>k<strong>in</strong>g <strong>water</strong>with pH values between 8.3 and 9.4. Corrosive alkal<strong>in</strong>ity (hydroxyl alkal<strong>in</strong>ity from sodiumhydroxide, NaOH) can only exist at pH values above 9.4. Furthermore, when <strong>water</strong> istreated for adjustment <strong>in</strong> pH the content <strong>of</strong> other components has to be quantified.23
Review <strong>of</strong> the literatureFigure 4. The distribution <strong>of</strong> carbonate species as a fraction <strong>of</strong> totaldissolved carbonate <strong>in</strong> relation to solution pH.3.3. Copper <strong>in</strong>take from dr<strong>in</strong>k<strong>in</strong>g <strong>water</strong>The daily consumption <strong>of</strong> copper through diet lies between 1 and 2 mg/day <strong>in</strong> adults and0.6 to 0.8 mg/day <strong>in</strong> two-year-old children. Copper is present <strong>in</strong> all foods but the highestconcentrations can be found <strong>in</strong> liver, nuts, seeds and chocolate (around 10 mg/kg). 159, 160Dr<strong>in</strong>k<strong>in</strong>g <strong>water</strong> is not normally <strong>in</strong>cluded <strong>in</strong> the daily <strong>in</strong>take <strong>of</strong> copper and the total <strong>in</strong>take <strong>of</strong>copper can vary greatly depend<strong>in</strong>g on the copper concentration <strong>in</strong> the dr<strong>in</strong>k<strong>in</strong>g <strong>water</strong>consumed.More than 90 percent <strong>of</strong> the <strong>water</strong> pipe system <strong>in</strong> the USA, Sweden, Great Brita<strong>in</strong> andNorway consists <strong>of</strong> copper pipes. The proportion <strong>of</strong> copper pipes <strong>in</strong> the <strong>water</strong> system is40-60 percent for Germany, Spa<strong>in</strong>, France and Italy but only 12 percent <strong>in</strong> Japan. 161 Asurvey <strong>in</strong> Seattle (USA) estimated that the <strong>in</strong>take <strong>of</strong> copper from dr<strong>in</strong>k<strong>in</strong>g <strong>water</strong> wasapproximately 10 times higher among persons with copper pipes (1.3-2.2 mg per day) <strong>in</strong>their homes than for those hav<strong>in</strong>g galvanized pipes. Thus, if copper pipes corrode, theconcentration <strong>of</strong> copper <strong>in</strong> dr<strong>in</strong>k<strong>in</strong>g <strong>water</strong> will rise. 162 The corrosion <strong>of</strong> copper pipes is<strong>in</strong>fluenced by the time dr<strong>in</strong>k<strong>in</strong>g <strong>water</strong> stays <strong>in</strong> the pipes. Flush<strong>in</strong>g the tap can reduce the24
- Page 1 and 2: Pro-oxidant activity of vitamin C i
- Page 3 and 4: Supervised byDocent Tommy Nordströ
- Page 5 and 6: ContentsCONTENTSLIST OF ORIGINAL PU
- Page 8 and 9: List of original publicationsLIST O
- Page 10 and 11: AcknowledgementsACKNOWLEDGEMENTSThi
- Page 12 and 13: AbbreviationsABBREVIATIONSAsc …
- Page 14 and 15: Review of the literatureREVIEW OF T
- Page 16 and 17: Review of the literatureSince vitam
- Page 18 and 19: Review of the literaturestill added
- Page 20 and 21: Review of the literatureantioxidant
- Page 22 and 23: Review of the literatureThe α-toco
- Page 24 and 25: Review of the literatureCopper, wil
- Page 26 and 27: Review of the literatureOH • + H
- Page 28 and 29: Review of the literatureFormation o
- Page 32 and 33: Review of the literaturecopper conc
- Page 34 and 35: Experimental proceduresEXPERIMENTAL
- Page 36 and 37: Experimental procedures2.2. Measure
- Page 38 and 39: Experimental procedurestetrahydrate
- Page 40 and 41: ResultsRESULTS1. Vitamin C induces
- Page 42 and 43: Results3. Oxidative decomposition o
- Page 44 and 45: Resultsdifferent water samples vari
- Page 46 and 47: DiscussionDISCUSSIONNowadays, ascor
- Page 48 and 49: DiscussionCu 2+ + Asc → Cu + + As
- Page 50 and 51: Discussion3. Iron inhibits vitamin
- Page 52 and 53: Discussionperoxide might have an im
- Page 54 and 55: ConclusionsCONCLUSIONSThe main focu
- Page 56 and 57: ReferencesREFERENCES1. Arrigoni O,
- Page 58 and 59: References34. Padayatty SJ, Katz A,
- Page 60 and 61: References66. Sies H, Stahl W, Sund
- Page 62 and 63: References95. Halliwell B. Role of
- Page 64 and 65: References127. Park S, Han SS, Park
- Page 66 and 67: References157. Critchley MM, Cromar
- Page 68 and 69: References185. Liao CH, Kang SF, Wu
- Page 70 and 71: References214. Orr CW. Studies on a
- Page 72: References243. Miller C, Kennington
- Page 88 and 89:
Free Radical Research, Volume 38 Nu
- Page 90 and 91:
VITAMIN C OXIDATION IN DRINKING WAT
- Page 92 and 93:
VITAMIN C OXIDATION IN DRINKING WAT
- Page 94 and 95:
Free Radical Research, May 2005; 39
- Page 96 and 97:
Iron inhibits Vitamin C/copper-indu
- Page 98 and 99:
Iron inhibits Vitamin C/copper-indu
- Page 100 and 101:
Free Radical Research, November 200
- Page 102 and 103:
Hydrogen peroxide formation in drin
- Page 104 and 105:
Hydrogen peroxide formation in drin
- Page 106 and 107:
Hydrogen peroxide formation in drin