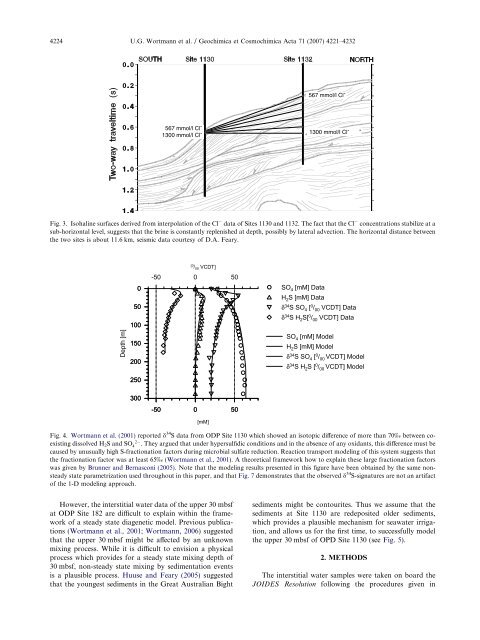
4224 U.G. Wortmann et al. / Geochimica et Cosmochimica Acta 71 (2007) 4221–4232567 mmol/l Cl - 567 mmol/l Cl -1300 mmol/l Cl - 1300 mmol/l Cl -Fig. 3. Isohal<strong>in</strong>e surfaces derived from <strong>in</strong>terpolation <strong>of</strong> <strong>the</strong> Cl data <strong>of</strong> Sites 1130 and 1132. The fact that <strong>the</strong> Cl concentrations stabilize at asub-horizontal level, suggests that <strong>the</strong> br<strong>in</strong>e is constantly replenished at depth, possibly by lateral advection. The horizontal distance between<strong>the</strong> two sites is about 11.6 km, seismic data courtesy <strong>of</strong> D.A. Feary.Depth [m]-50 0 500SO 4 [mM] DataH 2 S [mM] Data50δ 34 S SO 4 [ 0 / 00 VCDT] Dataδ 34 S H 2 S[ 0 / 00 VCDT] Data1001502002500 / 00[VCDT]SO 4 [mM] ModelH 2 S [mM] Modelδ 34 S SO 4 [ 0 / 00 VCDT] Modelδ 34 S H 2 S [ 0 / 00 VCDT] Model300-50 0 50[mM]Fig. 4. Wortmann et al. (2001) reported d 34 S data from ODP Site 1130 which showed an isotopic difference <strong>of</strong> more than 70‰ between coexist<strong>in</strong>gdissolved H 2 S and SO 24. They argued that under hypersulfidic conditions and <strong>in</strong> <strong>the</strong> absence <strong>of</strong> any oxidants, this difference must becaused by unusually high S-fractionation factors dur<strong>in</strong>g microbial <strong>sulfate</strong> reduction. Reaction transport model<strong>in</strong>g <strong>of</strong> this system suggests that<strong>the</strong> fractionation factor was at least 65‰ (Wortmann et al., 2001). A <strong>the</strong>oretical framework how to expla<strong>in</strong> <strong>the</strong>se large fractionation factorswas given by Brunner and Bernasconi (2005). Note that <strong>the</strong> model<strong>in</strong>g results presented <strong>in</strong> this figure have been obta<strong>in</strong>ed by <strong>the</strong> same nonsteadystate parametrization used throughout <strong>in</strong> this paper, and that Fig. 7 demonstrates that <strong>the</strong> observed d 34 S-signatures are not an artifact<strong>of</strong> <strong>the</strong> 1-D model<strong>in</strong>g approach.However, <strong>the</strong> <strong>in</strong>terstitial <strong>water</strong> data <strong>of</strong> <strong>the</strong> upper 30 mbsfat ODP Site 182 are difficult to expla<strong>in</strong> with<strong>in</strong> <strong>the</strong> framework<strong>of</strong> a steady state diagenetic model. Previous publications(Wortmann et al., 2001; Wortmann, 2006) suggestedthat <strong>the</strong> upper 30 mbsf might be affected by an unknownmix<strong>in</strong>g process. While it is difficult to envision a physicalprocess which provides for a steady state mix<strong>in</strong>g depth <strong>of</strong>30 mbsf, non-steady state mix<strong>in</strong>g by sedimentation eventsis a plausible process. Huuse and Feary (2005) suggestedthat <strong>the</strong> youngest sediments <strong>in</strong> <strong>the</strong> Great Australian Bightsediments might be contourites. Thus we assume that <strong>the</strong>sediments at Site 1130 are redeposited older sediments,which provides a plausible mechanism for sea<strong>water</strong> irrigation,and allows us for <strong>the</strong> first time, to successfully model<strong>the</strong> upper 30 mbsf <strong>of</strong> OPD Site 1130 (see Fig. 5).2. METHODSThe <strong>in</strong>terstitial <strong>water</strong> samples were taken on board <strong>the</strong>JOIDES Resolution follow<strong>in</strong>g <strong>the</strong> procedures given <strong>in</strong>
<strong>Oxygen</strong> <strong>isotope</strong> <strong>biogeochemistry</strong> <strong>of</strong> <strong>pore</strong> <strong>water</strong> <strong>sulfate</strong>... 4225Site 1130 14 C dataSite 1130 new age modelSite 1130 old age modelCl -New age modelOld age modelmbsf01020300 cm/y3.12 cm/y0 cm/y2.86 cm/y26 cm/kymbsf [m]010203040405010 0 10 1 10 2 10 3 10 4 10 5Age [yrs B.P.]50500 1.000 1.500Cl - [mM]Fig. 5. Comparison between <strong>the</strong> Site 1130 14 C age data, and <strong>the</strong> age model used <strong>in</strong> our model. We assume that <strong>the</strong> sediments at Site 1130 arereworked drift sediments (Huuse and Feary, 2005) and have been redeposited about 20–30 ky after <strong>the</strong>ir <strong>in</strong>itial deposition. Note that <strong>the</strong>model assumes two major re-sedimentation events (0.2–0.5 ky B.P. and 10.5–10.7 ky B.P.) and two prom<strong>in</strong>ent hiati (0–0.2 ky B.P, and 0.5–10.5 ky B.P.). The event and hiati tim<strong>in</strong>g and duration have been obta<strong>in</strong>ed from <strong>in</strong>verse model<strong>in</strong>g <strong>of</strong> <strong>the</strong> conservative Cl data <strong>of</strong> Site 1130,but are o<strong>the</strong>rwise unconstra<strong>in</strong>ed. 14 C age data from H<strong>in</strong>e et al. (2002).Feary et al. (2000b). Samples were treated immediatelyafter collection by add<strong>in</strong>g 100 lL <strong>of</strong> a saturated z<strong>in</strong>c acetatesolution to 5 mL <strong>of</strong> sample, to precipitate all H 2 S and <strong>in</strong>hibitfur<strong>the</strong>r activity <strong>of</strong> <strong>sulfate</strong>-reduc<strong>in</strong>g microorganisms. Theprecipitated ZnS was separated us<strong>in</strong>g a centrifuge, and <strong>the</strong>supernatant was filtered through a 0.45 lm membrane filterand acidified with HCl. The <strong>sulfate</strong> was precipitated asBaSO 4 by <strong>the</strong> addition <strong>of</strong> BaCl 2 with<strong>in</strong> one hour after acidification.Samples were centrifuged, washed with hot <strong>water</strong>,and dried. For d 18 O SO4 2 measurements, about 135 lgBaSO 4 was added to Ag cups and pyrolyzed at 1400 °Con a Hekatech HT-EA us<strong>in</strong>g helium as a carrier gas. Theproduced CO was routed through an Ascarite trap, separatedon a Molsieve 5A, and subsequently measured on aThermo F<strong>in</strong>nigan Mat 253 <strong>in</strong> cont<strong>in</strong>uous flow mode us<strong>in</strong>g<strong>the</strong> Conflo III open split <strong>in</strong>terface. The system was calibratedby us<strong>in</strong>g <strong>the</strong> <strong>in</strong>ternational standards USGS 32(25.7‰ V-SMOW) and NBS 127 (9.3‰ V-SMOW).IAEA-SO-6 was also measured ( 11.34‰ V-SMOW) butnot used for calibration purposes s<strong>in</strong>ce all data falls <strong>in</strong> <strong>the</strong>range between NBS 127 and USGS 32. Analytical reproducibility<strong>of</strong> <strong>the</strong> measurements was determ<strong>in</strong>ed by runn<strong>in</strong>gseveral replicates <strong>of</strong> an <strong>in</strong>-house standard (BaSO 4 withd 18 O = 12.38‰) with each run. We report <strong>the</strong> accumulatederror (1r) <strong>of</strong> <strong>in</strong> house standards measured at <strong>the</strong> beg<strong>in</strong>n<strong>in</strong>g,middle and end <strong>of</strong> each run from 12 <strong>in</strong>dividual runs (i.e., 3per run = 36 total) as ±0.26‰. The data are reported <strong>in</strong> <strong>the</strong>conventional delta notation with respect to V-SMOW.<strong>Oxygen</strong> isotopic measurements <strong>of</strong> H 2 O from <strong>in</strong>terstitial<strong>water</strong> samples were made <strong>in</strong> <strong>the</strong> Division <strong>of</strong> Mar<strong>in</strong>e Geologyand Geophysics at <strong>the</strong> University <strong>of</strong> Miami us<strong>in</strong>g a<strong>water</strong> equilibration system (WEST) attached to an EuropaGEO. This setup determ<strong>in</strong>es <strong>the</strong> oxygen isotopic compositionfrom CO 2 which has been <strong>in</strong>jected <strong>in</strong>to serum bottles(5 cm 3 ) at slightly above atmospheric pressure conta<strong>in</strong><strong>in</strong>g0.5 cm 3 <strong>of</strong> sample. The samples are subsequently equilibratedat 40 °C for 12 h without shak<strong>in</strong>g. The process isentirely automated with <strong>the</strong> CO 2 be<strong>in</strong>g <strong>in</strong>jected and retrievedus<strong>in</strong>g an autosampler and <strong>the</strong> gas be<strong>in</strong>g transferredto a dual-<strong>in</strong>let mass spectrometer through a cryogenic trap(at 70 °C) to remove <strong>water</strong>. The precision <strong>of</strong> this methodfor oxygen, determ<strong>in</strong>ed by measur<strong>in</strong>g 59 samples <strong>of</strong> our<strong>in</strong>ternal standard, is ±0.07‰. All data are reported <strong>in</strong>‰ relative to V-SMOW us<strong>in</strong>g <strong>the</strong> conventional deltanotation.The equilibrium values reported here are based on <strong>the</strong>experimental steady state values reported by Fritz et al.(1989), and we use <strong>the</strong> follow<strong>in</strong>g relation to calculate <strong>the</strong>equilibrium values reported <strong>in</strong> Fig. 8.d 18 O SO4 2 ¼ 29:772 T 0:1599 ð3Þwhere T denotes <strong>the</strong> shipboard measured temperature pr<strong>of</strong>ile<strong>of</strong> OPD Site 1130 (Feary et al., 2000a), which weapproximated asT ¼ 10:5 þ z 0:037ð4Þwhere z denotes <strong>the</strong> depth <strong>in</strong> meters below seafloor.Total iron <strong>of</strong> selected samples was measured by X-rayfluorescence spectroscopy (Philips PW 2400, equipped withRh-tube) on fused borate glass beads with a precision (2SD)<strong>of</strong> 1.1% (Dellwig et al., 2002). Total <strong>in</strong>organic reducible sulphur(TRIS) which is considered to essentially representpyrite sulfur, was extracted from <strong>the</strong> sediments accord<strong>in</strong>gto <strong>the</strong> one-step hot Cr(II)Cl 2 distillation method (Foss<strong>in</strong>gand Jørgensen, 1989) where <strong>the</strong> H 2 S was trapped quantitativelyas Ag 2 S <strong>in</strong> an AgNO 3 solution and quantified