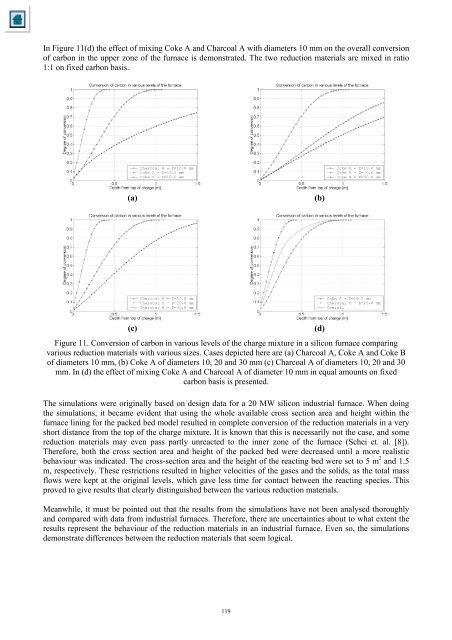
In Figure 11(d) <strong>the</strong> effect <strong>of</strong> mix<strong>in</strong>g <strong>Coke</strong> A <strong>and</strong> <strong>Char<strong>co</strong>al</strong> A with diameters 10 mm on <strong>the</strong> overall <strong>co</strong>nversion<strong>of</strong> carbon <strong>in</strong> <strong>the</strong> upper zone <strong>of</strong> <strong>the</strong> furnace is demonstrated. The two reduction materials are mixed <strong>in</strong> ratio1:1 on fixed carbon basis.(a)(b)(c)Figure 11. Conversion <strong>of</strong> carbon <strong>in</strong> various levels <strong>of</strong> <strong>the</strong> charge mixture <strong>in</strong> a sili<strong>co</strong>n furnace <strong>co</strong>mpar<strong>in</strong>gvarious reduction materials with various sizes. Cases depicted here are (a) <strong>Char<strong>co</strong>al</strong> A, <strong>Coke</strong> A <strong>and</strong> <strong>Coke</strong> B<strong>of</strong> diameters 10 mm, (b) <strong>Coke</strong> A <strong>of</strong> diameters 10, 20 <strong>and</strong> 30 mm (c) <strong>Char<strong>co</strong>al</strong> A <strong>of</strong> diameters 10, 20 <strong>and</strong> 30mm. In (d) <strong>the</strong> effect <strong>of</strong> mix<strong>in</strong>g <strong>Coke</strong> A <strong>and</strong> <strong>Char<strong>co</strong>al</strong> A <strong>of</strong> diameter 10 mm <strong>in</strong> equal amounts on fixedcarbon basis is presented.The simulations were orig<strong>in</strong>ally based on design data for a 20 MW sili<strong>co</strong>n <strong>in</strong>dustrial furnace. When do<strong>in</strong>g<strong>the</strong> simulations, it became evident that us<strong>in</strong>g <strong>the</strong> whole available cross section area <strong>and</strong> height with<strong>in</strong> <strong>the</strong>furnace l<strong>in</strong><strong>in</strong>g for <strong>the</strong> packed bed model resulted <strong>in</strong> <strong>co</strong>mplete <strong>co</strong>nversion <strong>of</strong> <strong>the</strong> reduction materials <strong>in</strong> a veryshort distance from <strong>the</strong> top <strong>of</strong> <strong>the</strong> charge mixture. It is known that this is necessarily not <strong>the</strong> case, <strong>and</strong> somereduction materials may even pass partly unreacted to <strong>the</strong> <strong>in</strong>ner zone <strong>of</strong> <strong>the</strong> furnace (Schei et. al. [8]).Therefore, both <strong>the</strong> cross section area <strong>and</strong> height <strong>of</strong> <strong>the</strong> packed bed were decreased until a more realisticbehaviour was <strong>in</strong>dicated. The cross-section area <strong>and</strong> <strong>the</strong> height <strong>of</strong> <strong>the</strong> react<strong>in</strong>g bed were set to 5 m 2 <strong>and</strong> 1.5m, respectively. These restrictions resulted <strong>in</strong> higher velocities <strong>of</strong> <strong>the</strong> gases <strong>and</strong> <strong>the</strong> solids, as <strong>the</strong> total massflows were kept at <strong>the</strong> orig<strong>in</strong>al levels, which gave less time for <strong>co</strong>ntact between <strong>the</strong> react<strong>in</strong>g species. Thisproved to give results that clearly dist<strong>in</strong>guished between <strong>the</strong> various reduction materials.Meanwhile, it must be po<strong>in</strong>ted out that <strong>the</strong> results from <strong>the</strong> simulations have not been analysed thoroughly<strong>and</strong> <strong>co</strong>mpared with data from <strong>in</strong>dustrial furnaces. Therefore, <strong>the</strong>re are uncerta<strong>in</strong>ties about to what extent <strong>the</strong>results represent <strong>the</strong> behaviour <strong>of</strong> <strong>the</strong> reduction materials <strong>in</strong> an <strong>in</strong>dustrial furnace. Even so, <strong>the</strong> simulationsdemonstrate differences between <strong>the</strong> reduction materials that seem logical.(d)
Some o<strong>the</strong>r simplifications <strong>and</strong> assumptions were also made to m<strong>in</strong>imise <strong>the</strong> number <strong>of</strong> parameters that<strong>co</strong>uld vary <strong>and</strong> affect <strong>the</strong> <strong>in</strong>terpretation <strong>of</strong> <strong>the</strong> simulated cases:• The flows <strong>of</strong> quartz <strong>and</strong> carbon were <strong>co</strong>nstant, with fixed carbon <strong>co</strong>verage <strong>of</strong> <strong>the</strong> charge mixture held at90 %.• The gas flow <strong>and</strong> gas <strong>co</strong>mposition enter<strong>in</strong>g <strong>the</strong> bed <strong>of</strong> reduction materials from <strong>in</strong>ner zones <strong>of</strong> <strong>the</strong>furnace were held <strong>co</strong>nstant for all cases, not tak<strong>in</strong>g <strong>in</strong>to ac<strong>co</strong>unt <strong>the</strong> total stoichiometry <strong>of</strong> <strong>the</strong> furnace• The temperature was held at 1650°C, as <strong>the</strong> shr<strong>in</strong>k<strong>in</strong>g <strong>co</strong>re model <strong>and</strong> <strong>the</strong> packed bed model areiso<strong>the</strong>rmal <strong>and</strong> <strong>the</strong> k<strong>in</strong>etic parameters were estimated with data from experiments at this temperature.7. CONCLUSIONSBased on laboratory experiments where <strong>the</strong> reaction k<strong>in</strong>etics between carbonaceous spheres <strong>and</strong> SiO-gaswere studied, estimation <strong>of</strong> k<strong>in</strong>etic parameters <strong>in</strong> a ma<strong>the</strong>matical model, which describes <strong>the</strong> degree <strong>of</strong><strong>co</strong>nversion versus time, was carried out. The shr<strong>in</strong>k<strong>in</strong>g <strong>co</strong>re model proved to give <strong>the</strong> best description <strong>of</strong> <strong>the</strong>overall reaction k<strong>in</strong>etics for <strong>the</strong> particles, but does not give a <strong>co</strong>mplete representative description <strong>of</strong> <strong>the</strong><strong>co</strong>nversion progress <strong>in</strong> <strong>the</strong> <strong>in</strong>ner <strong>of</strong> <strong>the</strong> particles. Some deviations between <strong>the</strong> shr<strong>in</strong>k<strong>in</strong>g <strong>co</strong>re model <strong>and</strong> <strong>the</strong>experimental data are observed. Most <strong>of</strong> <strong>the</strong>se deviations are probably due to <strong>the</strong> <strong>in</strong>homogeneous nature <strong>of</strong><strong>the</strong> carbonaceous materials.The estimated parameters were used <strong>in</strong> packed bed models with <strong>the</strong> shr<strong>in</strong>k<strong>in</strong>g <strong>co</strong>re model for <strong>the</strong> <strong>in</strong>dividualparticles to study <strong>the</strong>oretically how different reduction materials perform <strong>in</strong> <strong>the</strong> SINTEF SiO-reactivity test<strong>and</strong> <strong>in</strong> a sili<strong>co</strong>n furnace. Results from simulations <strong>and</strong> experiments with <strong>the</strong> SINTEF SiO-reactivity test were<strong>co</strong>mpared <strong>and</strong> agreed very well. This validated <strong>in</strong> high degree <strong>the</strong> chosen model <strong>and</strong> <strong>the</strong> estimatedparameters. In addition, <strong>the</strong> estimated parameters agreed with results from an earlier work.Similarly, based on <strong>the</strong> mentioned models <strong>and</strong> parameters, several cases were simulated for a hypo<strong>the</strong>ticalsili<strong>co</strong>n furnace with some selected reduction materials <strong>of</strong> various sizes <strong>and</strong> spann<strong>in</strong>g a range <strong>of</strong> SiOreactivityfigures. In order to achieve reasonable differences <strong>in</strong> responses for <strong>the</strong> selected reduction materials,<strong>the</strong> cross section area <strong>and</strong> height <strong>of</strong> <strong>the</strong> bed were assumed substantial smaller than <strong>the</strong> available space with<strong>in</strong><strong>the</strong> l<strong>in</strong><strong>in</strong>g <strong>of</strong> <strong>the</strong> furnace. The validity <strong>of</strong> <strong>the</strong>se assumptions is somewhat uncerta<strong>in</strong>, as <strong>the</strong> results have notbeen <strong>co</strong>mpared with data from <strong>in</strong>dustrial furnaces. However, <strong>the</strong> simulations <strong>in</strong>dicate some differencesbetween <strong>the</strong> reduction materials that appear logical.8. ACKNOWLEDGEMENTSThe authors are grateful to “The Biocarbon Project” governed by <strong>the</strong> Norwegian Ferro Alloy ResearchAssociation for <strong>co</strong>ntributions to this work <strong>and</strong> for <strong>the</strong> f<strong>in</strong>ancial support from <strong>the</strong> Norwegian ResearchCouncil.9. REFERENCES[1] Myrhaug, E. <strong>and</strong> Tveit, H., “Material balances <strong>of</strong> trace elements <strong>in</strong> <strong>the</strong> ferrosili<strong>co</strong>n <strong>and</strong> sili<strong>co</strong>nprocesses”, 58th Electric Furnace Conference, Orl<strong>and</strong>o, Florida, November 2000.[2] Tuset, J.Kr. <strong>and</strong> Raanes, O., “Reactivity <strong>of</strong> reduction materials <strong>in</strong> <strong>the</strong> production <strong>of</strong> sili<strong>co</strong>n, sili<strong>co</strong>n-richferroalloys <strong>and</strong> sili<strong>co</strong>n carbide”, AIME El. Furnace Conf., St. Louis, Miss., 7-10 Dec. 1976.[3] Paull, J.M. <strong>and</strong> See, J.B., “The <strong>in</strong>teraction <strong>of</strong> sili<strong>co</strong>n monoxide gas with carbonaceous reduc<strong>in</strong>g agents”,Journal <strong>of</strong> South African Institute <strong>of</strong> M<strong>in</strong><strong>in</strong>g <strong>and</strong> Metallurgy, vol. 79 (2), pp. 35-41, 1978.[4] Videm, Th., “<strong>Reaction</strong> rate <strong>of</strong> reduction materials for <strong>the</strong> (ferro)sili<strong>co</strong>n process”, INFACON 7,Trondheim, Norway, June 1995.[5] Raaness, O. <strong>and</strong> Gray, R., “Coal <strong>in</strong> <strong>the</strong> production <strong>of</strong> sili<strong>co</strong>n rich alloys”, INFACON 7, Trondheim,Norway, June 1995.[6] Myrhaug, E., “Non-fossil reduction materials <strong>in</strong> <strong>the</strong> sili<strong>co</strong>n process - properties <strong>and</strong> behaviour”, Dr. <strong>in</strong>g.<strong>the</strong>sis, Norwegian University <strong>of</strong> Science <strong>and</strong> Technology, Norway. To be submitted <strong>in</strong> <strong>the</strong> last part <strong>of</strong>2003.[7] Myrhaug, E. <strong>and</strong> Tuset, J.Kr., “<strong>Reaction</strong> K<strong>in</strong>etics <strong>of</strong> <strong>Char<strong>co</strong>al</strong> <strong>and</strong> <strong>Coke</strong> <strong>in</strong> <strong>the</strong> Sili<strong>co</strong>n Processes”,Sili<strong>co</strong>n for <strong>the</strong> Chemical Industry VI, Loen, Norway, 2002.