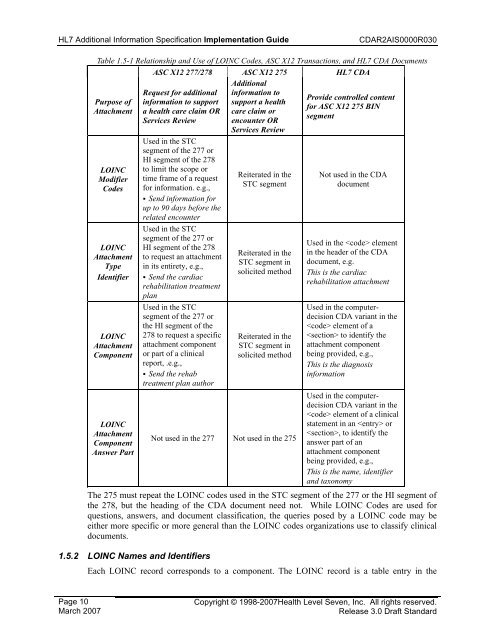
<strong>HL7</strong> Additional Information Specification <strong>Implementation</strong> <strong>Guide</strong><strong>CDA</strong><strong>R2</strong><strong>AIS</strong>0000R030Table 1.5-1 Relationship and Use of LOINC Codes, ASC X12 Transactions, and <strong>HL7</strong> <strong>CDA</strong> DocumentsASC X12 277/278 ASC X12 275 <strong>HL7</strong> <strong>CDA</strong>AdditionalRequest for additional information toProvide controlled contentPurpose of information to support support a healthfor ASC X12 275 BINAttachment a health care claim OR care claim orsegmentServices Review encounter ORServices ReviewLOINCModifierCodesLOINCAttachmentTypeIdentifierLOINCAttachmentComponentLOINCAttachmentComponentAnswer PartUsed in the STCsegment of the 277 orHI segment of the 278to limit the scope ortime frame of a requestfor information. e.g.,• Send information forup to 90 days before therelated encounterUsed in the STCsegment of the 277 orHI segment of the 278to request an attachmentin its entirety, e.g.,• Send the cardiacrehabilitation treatmentplanUsed in the STCsegment of the 277 orthe HI segment of the278 to request a specificattachment componentor part of a clinicalreport, .e.g.,• Send the rehabtreatment plan authorReiterated in theSTC segmentReiterated in theSTC segment insolicited methodReiterated in theSTC segment insolicited methodNot used in the 277 Not used in the 275Not used in the <strong>CDA</strong>documentUsed in the elementin the header of the <strong>CDA</strong>document, e.g.This is the cardiacrehabilitation attachmentUsed in the computerdecision<strong>CDA</strong> variant in the element of a to identify theattachment componentbeing provided, e.g.,This is the diagnosisinformationUsed in the computerdecision<strong>CDA</strong> variant in the element of a clinicalstatement in an or, to identify theanswer part of anattachment componentbeing provided, e.g.,This is the name, identifierand taxonomyThe 275 must repeat the LOINC codes used in the STC segment of the 277 or the HI segment ofthe 278, but the heading of the <strong>CDA</strong> document need not. While LOINC Codes are used forquestions, answers, and document classification, the queries posed by a LOINC code may beeither more specific or more general than the LOINC codes organizations use to classify clinicaldocuments.1.5.2 LOINC Names and IdentifiersEach LOINC record corresponds to a component. The LOINC record is a table entry in thePage 10March 2007Copyright © 1998-2007Health Level Seven, Inc. All rights reserved.Release 3.0 Draft Standard
<strong>HL7</strong> Additional Information Specification <strong>Implementation</strong> <strong>Guide</strong><strong>CDA</strong><strong>R2</strong><strong>AIS</strong>0000R030LOINC database maintained by the Regenstrief Institute and the LOINC Committee. See section1.5.4 for information on how to obtain the LOINC database. A LOINC record includes attributesto specify:• The numeric code that identifies the component,• The component name — e.g., potassium, hepatitis C antigen, distance the patient wastransported (by an ambulance)• The property reported — e.g., a mass concentration, length (distance)• The time aspect — e.g., Whether the measurement is a momentary observation at a pointin time, or an observation made over a span of time• The source of the data used in the reported information — e.g., urine, blood, EMStransport• The type of scale — e.g., whether the measurement is quantitative (a true measurement),nominal (red, blue, green), or simply narrative text providing the requested information• Where relevant, the method used to produce the result or other observation• A class code that associates the observation with others in a group, such as theobservations associated with an obstetric ultrasound procedureMany medical concepts have multiple LOINC codes. The codes distinguish different methods ofmaking the observation. For example, there are different LOINC codes for manual and automatedleukocyte counts. Indeed, there are three codes for the patient’s body weight according to whetherit was measured, estimated, or the datum is the weight that the patient reported.Different LOINC codes are also used to distinguish different ways to report the observation. Forexample, 10221-0 identifies the specimens taken during surgery when reported using narrativetext, whereas 8721-3 would identify coded descriptions of the same specimens.LOINC codes may also identify sets of observations. For example, the LOINC code 18674-2(ALCOHOL-SUBSTANCE ABUSE REHABILITATION TREATMENT PLAN, LONGESTPERIOD OF SOBRIETY FOR ABUSED SUBSTANCE) identifies a set of other observations,identified by other LOINC codes, including 18676-7 (ALCOHOL-SUBSTANCE ABUSEREHABILITATION TREATMENT PLAN, LONGEST PERIOD OF SOBRIETY), and 18675-9(ALCOHOL-SUBSTANCE ABUSE REHABILITATION TREATMENT PLAN, ABUSEDSUBSTANCE).The LOINC codes are not intended to transmit all possible information about a test orobservation. They are only intended to identify the observation. The LOINC code for a name isunique and permanent. The LOINC code has no intrinsic structure except that the last character inthe code is a mod-10 check digit.LOINC codes must always be transmitted without leading zeroes and with a hyphen beforethe check digit (e.g., "8709-8" and "10154-3").The LOINC Committee assigns LOINC codes upon request from various agencies. In the contextof attachments, the LOINC codes are obtained by the <strong>HL7</strong> ASIG. The ASIG forwards appropriaterequests to the LOINC committee for consideration. Requests go through a review process toensure the concept has not been previously added and the meaning is clear.Copyright © 1998-2007 Health Level Seven, Inc. All rights reserved.Release 3.0 Draft StandardPage 11March 2007