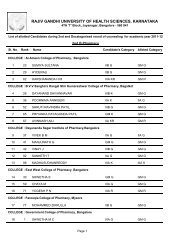
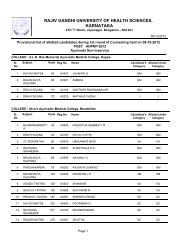
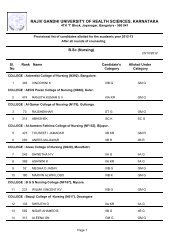
26II. Identification <strong>of</strong> organic compounds belonging to the following classes by :Systematic qualitative organic analysis including preparation <strong>of</strong> derivativesPhenols, amides, carbohydrates, amines, carboxylic acids, aldehyde and ketones,Alcohols, esters, hydrocarbons, anilides, nitrocompounds.III. Introduction to the use <strong>of</strong> stereo models:Methane, Ethane, Ethylene, Acetylene, Cis alkene, Trans alkene, inversion <strong>of</strong>configuration.Scheme <strong>of</strong> Practical Examination:SessionalsAnnualSynopsis 05 15Major Experiment 10 25Minor Experiment 03 15Viva 02 15Max Marks 20 70Duration 03hrs 04hrsNote : Total sessional marks is 30 (20 for practical sessional plus 10 marks forregularity, promptness, viva-voce and record maintenance).
271.5 PHARMACEUTICAL INORGANIC CHEMISTRY (THEORY)Theory : 2 Hrs./Week1. Scope and objectives: This course mainly deals with fundamentals <strong>of</strong>Analytical chemistry and also the study <strong>of</strong> inorganic pharmaceuticals regardingtheir monographs and also the course deals with basic knowledge <strong>of</strong>analysis <strong>of</strong> various pharmaceuticals.2. Upon completion <strong>of</strong> the course student shall be able to:a. under stand the principles and procedures <strong>of</strong> analysis <strong>of</strong> drugs and alsoregarding the application <strong>of</strong> inorganic pharmaceuticals;b. know the analysis <strong>of</strong> the inorganic pharmaceuticals their applications; andc. appreciate the importance <strong>of</strong> inorganic pharmaceuticals in preventing andcuring the disease.3. Coursematerials:Text booksa. A text book Inorganic medicinal chemistry by Surendra N. Pandeyab. A. H. Beckett and J. B. Stanlake’s Practical <strong>Pharm</strong>aceutical chemistry Vol -I& Vol-IIc. Inorganic <strong>Pharm</strong>aceutical Chemistry III-Edition P.Gundu RaoReference booksa. Inorganic <strong>Pharm</strong>aceutical Chemistry by Anand &Chetwal b. <strong>Pharm</strong>aceutical Inorganic chemistry byDr.B.G.Nagavic. Analytical chemistry principles by John H. Kennedyd. I.P.1985 and 1996, Govt. <strong>of</strong> India, Ministry <strong>of</strong> health4. Lecture wiseprogramme: Topics1 Errors: Errors in quantitative analysis, classification <strong>of</strong> errors, concept <strong>of</strong>accuracy and precision, treatment <strong>of</strong> analytical2 Volumetric analysis: Principle <strong>of</strong> volumetric analysis, different methods <strong>of</strong>analysis, different methods <strong>of</strong> expressing concentrations <strong>of</strong> solutions, Primary andsecondary standards.3 Acid-base titrations: Acid-base concepts, relative strength <strong>of</strong> acids and bases,law <strong>of</strong> mass action, common ion effect, ionic product <strong>of</strong> water, Henderson-Hasselbalch equation, buffer solutions, theory <strong>of</strong> indicators, neutralizationcurves, choice <strong>of</strong> indicators, mixed and universal indicators.4 Redox titrations: Concepts <strong>of</strong> oxidation and reduction reactions. Redoxreactions, theory <strong>of</strong> redox titrations, redox indicators, iodometry, iodimetry,titrations involving cerric sulphate, potassium iodate, potassium bromate, titanouschloride, Potassium permanganate.5 Non aqueous titrations: Theoretical basis, types <strong>of</strong> solvents, preparations andstandardization <strong>of</strong> titrant solutions, titration <strong>of</strong> weak acid, weak bases andindicators. Standardization <strong>of</strong> perchloric acid, lithium and sodium methoxide, tetrabutyl ammonium hydroxide.