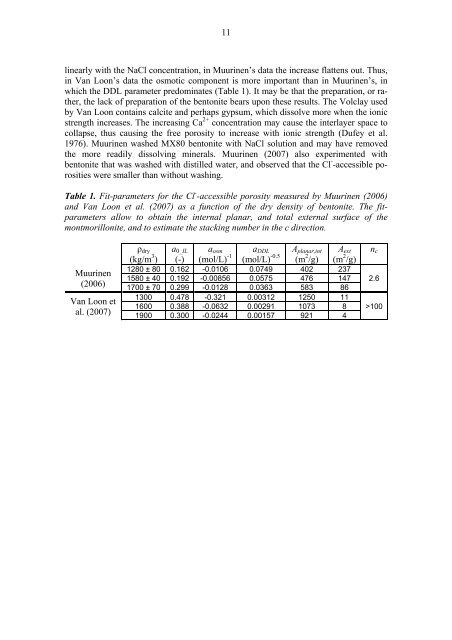
11l<strong>in</strong>early with the NaCl concentration, <strong>in</strong> Muur<strong>in</strong>en’s data the <strong>in</strong>crease flattens out. Thus,<strong>in</strong> Van Loon’s data the osmotic component is more important than <strong>in</strong> Muur<strong>in</strong>en’s, <strong>in</strong>which the DDL parameter predom<strong>in</strong>ates (Table 1). It may be that the preparation, or rather,the lack <strong>of</strong> preparation <strong>of</strong> the bentonite bears upon these results. The Volclay usedby Van Loon conta<strong>in</strong>s calcite <strong>and</strong> perhaps gypsum, which dissolve more when the ionicstrength <strong>in</strong>creases. The <strong>in</strong>creas<strong>in</strong>g Ca 2+ concentration may cause the <strong>in</strong>terlayer space tocollapse, thus caus<strong>in</strong>g the free <strong>porosity</strong> to <strong>in</strong>crease with ionic strength (Dufey et al.1976). Muur<strong>in</strong>en washed MX80 bentonite with NaCl solution <strong>and</strong> may have removedthe more readily dissolv<strong>in</strong>g m<strong>in</strong>erals. Muur<strong>in</strong>en (2007) also experimented withbentonite that was washed with distilled water, <strong>and</strong> observed that the Cl - -accessible porositieswere smaller than without wash<strong>in</strong>g.Table 1. Fit-parameters for the Cl - -accessible <strong>porosity</strong> measured by Muur<strong>in</strong>en (2006)<strong>and</strong> Van Loon et al. (2007) as a function <strong>of</strong> the dry density <strong>of</strong> bentonite. The fitparametersallow to obta<strong>in</strong> the <strong>in</strong>ternal planar, <strong>and</strong> total external surface <strong>of</strong> themontmorillonite, <strong>and</strong> to estimate the stack<strong>in</strong>g number <strong>in</strong> the c direction.Muur<strong>in</strong>en(2006)Van Loon etal. (2007)ρ dry(kg/m 3 )a 0_IL(-)a osm(mol/L) -1a DDL(mol/L) -0.5A planar,<strong>in</strong>t(m 2 /g)A ext(m 2 /g)1280 ± 80 0.162 -0.0106 0.0749 402 2371580 ± 40 0.192 -0.00856 0.0575 476 1471700 ± 70 0.299 -0.0128 0.0363 583 861300 0.478 -0.321 0.00312 1250 111600 0.388 -0.0632 0.00291 1073 81900 0.300 -0.0244 0.00157 921 4n c2.6>100
123 DIFFUSION IN BENTONITE: INTRODUCTIONThe diffusional flux <strong>of</strong> species i is given by:Jiuicii | z | F xi(14)where J i is the flux (mol/m 2 /s), u i is the mobility (m 2 /s/V), c i is the concentration(mol/m 3 ), z i is charge number (-), F is the Faraday constant (96485 J/V/eq), x is distance(m), <strong>and</strong> μ i is the thermodynamic potential, given by:μ i = μ i 0 + RT ln a i + z i Fψ (15)where μ i 0 is the st<strong>and</strong>ard potential (J/mol), R is the gas constant (8.314 J/K/mol), T isthe absolute temperature (K), a i is the activity (-), <strong>and</strong> ψ is the electrical potential (V).The activity is related to concentration by a i = γ i c i /c 0 , where γ i is the activity coefficient(-) <strong>and</strong> c i 0 is the st<strong>and</strong>ard state (1 mol/kg H 2 O, assumed equal to 1 mol/L <strong>in</strong> the follow<strong>in</strong>g).Assum<strong>in</strong>g an activity coefficient γ i = 1 <strong>and</strong> an electric potential gradient ∂ψ/∂x = 0, <strong>and</strong>us<strong>in</strong>g the identities u i = D w, i |z i | F / (RT) <strong>and</strong> c d(ln c) = d(c), Equation (14) becomes:Jidci Dw,i(16)dxwhere D w,i is the diffusion coefficient <strong>in</strong> water (m 2 /s) <strong>and</strong> c i has units mol/m 3 . In a porousmedium, the porewater diffusion coefficient differs from the diffusion coefficient<strong>in</strong> free water (D w,i ) by tortuosity <strong>and</strong> constrictivity factors (Van Brakel <strong>and</strong> Heertjes,1974; Figure 5):D p ,i D2 w,i(17)The tortuosity factor (θ 2 ) expresses that diffus<strong>in</strong>g molecules have to pass around solidgra<strong>in</strong>s <strong>and</strong> take a longer path (L a ) than the straight l<strong>in</strong>e distance (L) (Figure 5). A tortuouspath at an angle <strong>of</strong> 45° with the straight l<strong>in</strong>e distance obviously gives a tortuosity θ= 2 / √2 = 1.4, <strong>and</strong> a tortuosity factor θ 2 = 2. The constrictivity factor (δ) encompasseseffects <strong>of</strong> pore narrow<strong>in</strong>g <strong>and</strong> widen<strong>in</strong>g. A straight pore has a constrictivity factor <strong>of</strong> 1,if the pore narrows it becomes smaller than 1, <strong>and</strong> if it widens, larger than 1. Inert porousmedia have constrictivity factors slightly smaller than 1 (Van Brakel <strong>and</strong> Heertjes1974). However, <strong>in</strong> bentonite where DDL water occupies part <strong>of</strong> the pore space, porenarrow<strong>in</strong>g that results <strong>in</strong> overlapp<strong>in</strong>g DDL’s has a different effect for cations <strong>and</strong> neutralspecies that still can pass through, <strong>and</strong> for anions for which the constriction becomesan obstruction that has to be circumnavigated. In that case the constrictivity issmaller for anions than for cations <strong>and</strong> neutral species, while at the same time, for anionsthe tortuosity <strong>in</strong>creases.
- Page 1 and 2: Working Report 2013-29A Review of P
- Page 3 and 4: 2Katsaus bentoniitin huokoisuuteen
- Page 5 and 6: 21 INTRODUCTIONBentonite is foresee
- Page 7 and 8: 42 POROSITY IN BENTONITELaboratory
- Page 9 and 10: 6t IL = 1.41×10 -9 - 4.9×10 -13
- Page 11 and 12: 8data are few and not very precise
- Page 13: 10Figure 4. (A): Anion-accessible p
- Page 17 and 18: 14Dp,iDa,i (22)Rwhere R is the reta
- Page 19 and 20: 16In Equation (26) it is assumed th
- Page 21 and 22: 18Figure 7. The effective diffusion
- Page 23 and 24: 20The equivalent fraction of an exc
- Page 25 and 26: 22Table 3. Concentrations, concentr
- Page 27 and 28: 24Table 4. Specific volume and rela
- Page 29 and 30: 26Furthermore, if the flux of Cs +
- Page 31 and 32: 286 ACKNOWLEDGMENTSComments of Urs
- Page 33 and 34: 30Doherty, J., 1994. PEST, Model-in
- Page 35 and 36: 32Muurinen, A., Karnland, O. and Le