K. Guil<strong>in</strong>i et al. / Progress <strong>in</strong> Oceanography 110 (2013) 80–92 81<strong>and</strong> experimentally simulated depositions of phytodetritus is onlym<strong>in</strong>imal or absent compared to bacteria, foram<strong>in</strong>iferans <strong>and</strong> severalmacrofaunal taxa (e.g. Gooday, 1988; Graf, 1992; Pfannkuche,1993; Soltwedel, 1997; Galéron et al., 2001; Guil<strong>in</strong>i et al., 2010 <strong>and</strong>references <strong>the</strong>re<strong>in</strong>). The only rapid responses found to an episodicfood supply <strong>in</strong> <strong>the</strong> deep sea were an <strong>in</strong>crease <strong>in</strong> mean nematodesize at <strong>the</strong> German BIOTRANS site (April–July; Soltwedel et al.,1996) <strong>and</strong> nematode migration towards <strong>the</strong> sediment surface <strong>in</strong><strong>the</strong> Polar Front (December–January; Veit-Köhler et al., 2011). Doubledmeiofaunal abundance (ma<strong>in</strong>ly nematodes), from summer toautumn <strong>in</strong> sediments from <strong>the</strong> bathyal Mediterranean (de Bovéeet al., 1990) <strong>and</strong> <strong>the</strong> Nor<strong>the</strong>ast Atlantic along <strong>the</strong> Hebridean marg<strong>in</strong>(Mitchell et al., 1997) <strong>in</strong>dicate, however, a response on a longertime scale. Therefore, it is speculated that deep-sea nematodesare feed<strong>in</strong>g additionally or alternatively on ra<strong>the</strong>r unlimited, o<strong>the</strong>rfood sources than phytodetritus (e.g. bacteria, ciliates, flagellates,foram<strong>in</strong>iferans, fungi, DOC) <strong>and</strong> that <strong>the</strong>y may be characterizedby generally slower rates of colonization, respiration <strong>and</strong> somaticgrowth (Guil<strong>in</strong>i et al., 2010, 2011). Rigid evidence that any of <strong>the</strong>sefood sources are of substantial importance to ma<strong>in</strong>ta<strong>in</strong> <strong>the</strong> metabolicdem<strong>and</strong>s of nematodes, or respiration measurements under<strong>in</strong> situ conditions are, however, lack<strong>in</strong>g.This study describes <strong>and</strong> comb<strong>in</strong>es both structural <strong>and</strong> functionalaspects of deep-sea nematode assemblages along a latitud<strong>in</strong>altransect follow<strong>in</strong>g <strong>the</strong> Prime Meridian across <strong>the</strong> SO (49–70°S).Besides a spatial scale, a <strong>temporal</strong> scale was considered with regardsto <strong>the</strong> short-term response of nematode assemblages to aseasonal deposition of particulate organic matter (POM). Therefore,a station located <strong>in</strong> <strong>the</strong> Polar Front (52°2 0 S, 0°1 0 W), where a phytoplanktonbloom had settled dur<strong>in</strong>g <strong>the</strong> course of <strong>the</strong> expedition,was revisited after 52 days to perform repeated measurements.Overall, structural aspects comprise st<strong>and</strong><strong>in</strong>g stocks <strong>and</strong> <strong>community</strong>composition, while functional aspects refer to feed<strong>in</strong>g ecology,which is <strong>in</strong>vestigated by means of fatty acid (FA) compositions.Nematode FA compositions are determ<strong>in</strong>ed to provide a time-<strong>in</strong>tegratedview on <strong>the</strong> organisms’ diets at <strong>the</strong> <strong>community</strong> level. The FAmarker concept relies on <strong>the</strong> fact that certa<strong>in</strong> FA are <strong>in</strong>corporated<strong>in</strong>to consumers <strong>in</strong> a conservative manner (e.g. Dalsgaard et al.,2003; Lee et al., 2006). By compar<strong>in</strong>g nematode FA with what isknown from literature on surface water POM, o<strong>the</strong>r benthic organisms,bulk sediment <strong>and</strong> bottom-water POM properties, we aim toprovide <strong>in</strong>sights as to <strong>the</strong> resources used by deep-sea nematodecommunities across <strong>the</strong> Sou<strong>the</strong>rn Ocean. Although this techniqueis widely spread <strong>in</strong> mar<strong>in</strong>e ecological studies (see review by Kelly<strong>and</strong> Scheibl<strong>in</strong>g (2012)), it was only applied three times on mar<strong>in</strong>efree-liv<strong>in</strong>g nematodes (Leduc, 2009; Leduc <strong>and</strong> Probert, 2009; VanGaever et al., 2009).The follow<strong>in</strong>g hypo<strong>the</strong>ses were tested:(1) Structural <strong>and</strong> functional characteristics of deep-sea nematodeassemblages do not differ along a latitud<strong>in</strong>al gradientacross <strong>the</strong> SO.(2) Deep-sea nematodes at <strong>the</strong> Polar Front do not respond to aseasonal pulse of organic matter to <strong>the</strong> seafloor.2. Materials <strong>and</strong> methods2.1. Study site <strong>and</strong> sampl<strong>in</strong>g procedureSamples were collected dur<strong>in</strong>g <strong>the</strong> ANDEEP-SYSTCO expeditionon board of <strong>the</strong> RV Polarstern (ANT XXIV/2, 28.11.2007–04.02.2008). Sediment samples were taken at depths from1935 to 5323 m, at six sites along a north–south gradient, <strong>in</strong>close proximity of <strong>the</strong> Prime Meridian between 49°S <strong>and</strong> 70°S;across <strong>the</strong> Polar Front, <strong>the</strong> Weddell Sea <strong>and</strong> <strong>the</strong> Lazarev Sea(Fig. 1, Table 1). The stations are fur<strong>the</strong>r <strong>in</strong>dicated with <strong>the</strong>irabbreviations (PF: Polar Front, sPF: south Polar Front, cWS: centralWeddell Sea, MR: Maud Rise, LS: Lazarev Sea). All stationsoccur <strong>in</strong> different water masses <strong>and</strong> environments with differenttopographical, sedimentary <strong>and</strong> productivity conditions. The PF,sPF <strong>and</strong> cWS station are situated on <strong>the</strong> abyssal sea floor, MRis a seamount <strong>and</strong> LS is located at <strong>the</strong> cont<strong>in</strong>ental slope. The stationlocated at a sou<strong>the</strong>rn position <strong>in</strong> <strong>the</strong> Polar Front (sPF, at52°S) was visited twice; once when a phytoplankton bloomwas detected <strong>in</strong> <strong>the</strong> euphotic water layer based on <strong>in</strong>creasedfluorescence levels (06.12.2007; Herrmann <strong>and</strong> Bathmann,2010) <strong>and</strong> 52 days later when <strong>the</strong> rema<strong>in</strong>s of <strong>the</strong> bloom had settledto <strong>the</strong> sea floor (26–27.01.2008). These rema<strong>in</strong>s were visibleas a greenish fluff layer on <strong>the</strong> sediment surface (Veit-Köhleret al., 2011). Sachs et al. (2009) calculated <strong>the</strong> labile portion of<strong>the</strong> organic carbon flux (LC org ) based on <strong>in</strong> situ <strong>and</strong> ex situ measurementsof O 2 profiles <strong>in</strong> surface sediments (Table 1).All meiobenthic samples were taken with a multiple corer(MUC) sampl<strong>in</strong>g device equipped with 12 plexiglass cores (<strong>in</strong>nerdiameter: 9.4 cm, equivalent to 69.4 cm 2 ). The MUC was deployedone to three times per station, depend<strong>in</strong>g on ship time availability<strong>and</strong> sea state. The processed samples were thus a comb<strong>in</strong>ation oftrue replicates <strong>and</strong> pseudo-replicates, s<strong>in</strong>ce different cores from<strong>the</strong> same MUC deployment do not meet <strong>the</strong> criteria of r<strong>and</strong>omsampl<strong>in</strong>g (Hurlbert, 1984). An overview of <strong>the</strong> processed samplesis given <strong>in</strong> Table 1. The s<strong>in</strong>gle MUC deployment at <strong>the</strong> Polar Frontstation was only partially successful. It was visually determ<strong>in</strong>edthat some cores were disturbed. The most undisturbed cores werereserved for FA analysis, <strong>in</strong> order to ensure enough organismscould be collected. From all stations, <strong>the</strong> sediment cores dest<strong>in</strong>edfor meiofauna <strong>community</strong> analysis were sliced <strong>in</strong>to 1-cm fractionsdown to 5 cm <strong>and</strong> toge<strong>the</strong>r with <strong>the</strong> supernatant water preserved<strong>in</strong> a borax-buffered 4% formaldehyde-seawater solution. Fur<strong>the</strong>rmore,a m<strong>in</strong>imum of four cores per deployment were used to scoopoff <strong>the</strong> upper 5 cm of sediment. This material was immediatelysieved <strong>in</strong> <strong>the</strong> lab with filtered seawater (32 lm mesh) over stackedsieves (1 mm, 500 lm, 100 lm <strong>and</strong> 32 lm) to prevent clogg<strong>in</strong>g.The reta<strong>in</strong>ed 500 lm, 100 lm <strong>and</strong> 32 lm fractions were pooled<strong>and</strong> stored at 80 °C. Back at <strong>the</strong> laboratory, <strong>the</strong> nematodes wereextracted for fatty acid analyses.2.2. Nematode parametersSediment samples dest<strong>in</strong>ed for identify<strong>in</strong>g <strong>the</strong> nematode <strong>community</strong>were r<strong>in</strong>sed with tap water over a 32 lm mesh sieve (noupper sieve size used). The fraction reta<strong>in</strong>ed on <strong>the</strong> 32 lm sievewas three times centrifugated with <strong>the</strong> colloidal silica polymerLevasil Ò (H.C. Stark, 200/40%, q = 1.17, for 6 m<strong>in</strong> at 4000 rpm)<strong>and</strong> kaol<strong>in</strong> (McIntyre <strong>and</strong> Warwick, 1984) to extract all organisms.After sta<strong>in</strong><strong>in</strong>g with Rose Bengal, all metazoan organisms weremanually sorted to higher taxon level (follow<strong>in</strong>g Higg<strong>in</strong>s <strong>and</strong> Thiel,1988) <strong>and</strong> counted under a Leica MZ 12.5 stereomicroscope (8–100 magnification). Where possible, about 50–100 nematodeswere picked out r<strong>and</strong>omly with a f<strong>in</strong>e needle from each centimetersediment layer. They were gradually transferred to glycer<strong>in</strong>e (Se<strong>in</strong>horst,1959) before be<strong>in</strong>g mounted on glass slides. Nematodeswere identified to genus level under a compound microscope(1000 magnification). Adults were dist<strong>in</strong>guished from juvenilesbased on <strong>the</strong> development of a vulva <strong>and</strong> uterus <strong>in</strong> females <strong>and</strong> agonad <strong>and</strong> spicules <strong>in</strong> males. Based on mouth morphology, all identified<strong>in</strong>dividuals were classified <strong>in</strong>to four feed<strong>in</strong>g type groupsaccord<strong>in</strong>g to Wieser (1953): selective deposit feeders (1A), nonselectivedeposit feeders (1B), epistratum feeders (2A), <strong>and</strong> predators/scavengers(2B). By us<strong>in</strong>g a Leica DMR compound microscope<strong>and</strong> Leica LAS 3.3 imag<strong>in</strong>g software, nematode length (L, filiformtail excluded) <strong>and</strong> maximal width (W) were measured. Nematode
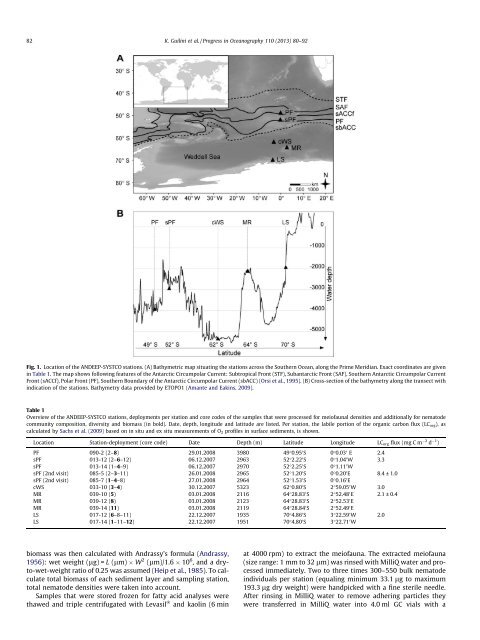
82 K. Guil<strong>in</strong>i et al. / Progress <strong>in</strong> Oceanography 110 (2013) 80–92Fig. 1. Location of <strong>the</strong> ANDEEP-SYSTCO stations. (A) Bathymetric map situat<strong>in</strong>g <strong>the</strong> stations across <strong>the</strong> Sou<strong>the</strong>rn Ocean, along <strong>the</strong> Prime Meridian. Exact coord<strong>in</strong>ates are given<strong>in</strong> Table 1. The map shows follow<strong>in</strong>g features of <strong>the</strong> Antarctic Circumpolar Current: Subtropical Front (STF), Subantarctic Front (SAF), Sou<strong>the</strong>rn Antarctic Circumpolar CurrentFront (sACCf), Polar Front (PF), Sou<strong>the</strong>rn Boundary of <strong>the</strong> Antarctic Circumpolar Current (sbACC) (Orsi et al., 1995). (B) Cross-section of <strong>the</strong> bathymetry along <strong>the</strong> transect with<strong>in</strong>dication of <strong>the</strong> stations. Bathymetry data provided by ETOPO1 (Amante <strong>and</strong> Eak<strong>in</strong>s, 2009).Table 1Overview of <strong>the</strong> ANDEEP-SYSTCO stations, deployments per station <strong>and</strong> core codes of <strong>the</strong> samples that were processed for meiofaunal densities <strong>and</strong> additionally for nematode<strong>community</strong> composition, diversity <strong>and</strong> biomass (<strong>in</strong> bold). Date, depth, longitude <strong>and</strong> latitude are listed. Per station, <strong>the</strong> labile portion of <strong>the</strong> organic carbon flux (LC org ), ascalculated by Sachs et al. (2009) based on <strong>in</strong> situ <strong>and</strong> ex situ measurements of O 2 profiles <strong>in</strong> surface sediments, is shown.Location Station-deployment (core code) Date Depth (m) Latitude Longitude LC org flux (mg C m 2 d 1 )PF 090-2 (2–8) 29.01.2008 3980 49°0.95 0 S 0°0.03 0 E 2.4sPF 013-12 (2–6–12) 06.12.2007 2963 52°2.22 0 S 0°1.04 0 W 3.3sPF 013-14 (1–4–9) 06.12.2007 2970 52°2.25 0 S 0°1.11 0 WsPF (2nd visit) 085-5 (2–3–11) 26.01.2008 2965 52°1.20 0 S 0°0.20 0 E 8.4 ± 1.0sPF (2nd visit) 085-7 (1–4–8) 27.01.2008 2964 52°1.53 0 S 0°0.16 0 EcWS 033-10 (3–4) 30.12.2007 5323 62°0.80 0 S 2°59.05 0 W 3.0MR 039-10 (5) 03.01.2008 2116 64°28.83 0 S 2°52.48 0 E 2.1 ± 0.4MR 039-12 (8) 03.01.2008 2123 64°28.83 0 S 2°52.53 0 EMR 039-14 (11) 03.01.2008 2119 64°28.84 0 S 2°52.49 0 ELS 017-12 (6–8–11) 22.12.2007 1935 70°4.86 0 S 3°22.59 0 W 2.0LS 017-14 (1–11–12) 22.12.2007 1951 70°4.80 0 S 3°22.71 0 Wbiomass was <strong>the</strong>n calculated with Andrassy’s formula (Andrassy,1956): wet weight (lg) = L (lm) W 2 (lm)/1.6 10 6 , <strong>and</strong> a dryto-wet-weightratio of 0.25 was assumed (Heip et al., 1985). To calculatetotal biomass of each sediment layer <strong>and</strong> sampl<strong>in</strong>g station,total nematode densities were taken <strong>in</strong>to account.Samples that were stored frozen for fatty acid analyses werethawed <strong>and</strong> triple centrifugated with Levasil Ò <strong>and</strong> kaol<strong>in</strong> (6 m<strong>in</strong>at 4000 rpm) to extract <strong>the</strong> meiofauna. The extracted meiofauna(size range: 1 mm to 32 lm) was r<strong>in</strong>sed with MilliQ water <strong>and</strong> processedimmediately. Two to three times 300–550 bulk nematode<strong>in</strong>dividuals per station (equal<strong>in</strong>g m<strong>in</strong>imum 33.1 lg to maximum193.3 lg dry weight) were h<strong>and</strong>picked with a f<strong>in</strong>e sterile needle.After r<strong>in</strong>s<strong>in</strong>g <strong>in</strong> MilliQ water to remove adher<strong>in</strong>g particles <strong>the</strong>ywere transferred <strong>in</strong> MilliQ water <strong>in</strong>to 4.0 ml GC vials with a