Brochure: Carbon Additives for Polymer Compounds - Timcal Graphite
Brochure: Carbon Additives for Polymer Compounds - Timcal Graphite
Brochure: Carbon Additives for Polymer Compounds - Timcal Graphite
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
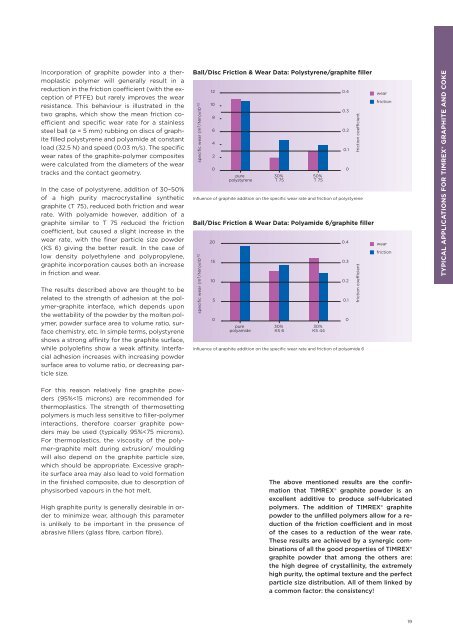
Incorporation of graphite powder into a thermoplastic<br />
polymer will generally result in a<br />
reduction in the friction coeffi cient (with the exception<br />
of PTFE) but rarely improves the wear<br />
resistance. This behaviour is illustrated in the<br />
two graphs, which show the mean friction coeffi<br />
cient and specifi c wear rate <strong>for</strong> a stainless<br />
steel ball (ø = 5 mm) rubbing on discs of graphite<br />
fi lled polystyrene and polyamide at constant<br />
load (32.5 N) and speed (0.03 m/s). The specifi c<br />
wear rates of the graphite-polymer composites<br />
were calculated from the diameters of the wear<br />
tracks and the contact geometry.<br />
In the case of polystyrene, addition of 30–50%<br />
of a high purity macrocrystalline synthetic<br />
graphite (T 75), reduced both friction and wear<br />
rate. With polyamide however, addition of a<br />
graphite similar to T 75 reduced the friction<br />
coeffi cient, but caused a slight increase in the<br />
wear rate, with the fi ner particle size powder<br />
(KS 6) giving the better result. In the case of<br />
low density polyethylene and polypropylene,<br />
graphite incorporation causes both an increase<br />
in friction and wear.<br />
The results described above are thought to be<br />
related to the strength of adhesion at the polymer-graphite<br />
interface, which depends upon<br />
the wettability of the powder by the molten polymer,<br />
powder surface area to volume ratio, surface<br />
chemistry, etc. In simple terms, polystyrene<br />
shows a strong affi nity <strong>for</strong> the graphite surface,<br />
while polyolefi ns show a weak affi nity. Interfacial<br />
adhesion increases with increasing powder<br />
surface area to volume ratio, or decreasing particle<br />
size.<br />
For this reason relatively fi ne graphite powders<br />
(95%