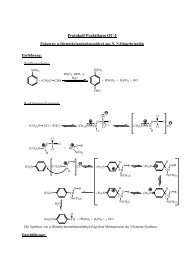
Gas-Phase Reactions of Homo- and ... - Institut für Chemie
Gas-Phase Reactions of Homo- and ... - Institut für Chemie
Gas-Phase Reactions of Homo- and ... - Institut für Chemie
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
1410<br />
Helvetica Chimica Acta ± Vol. 88 (2005)<br />
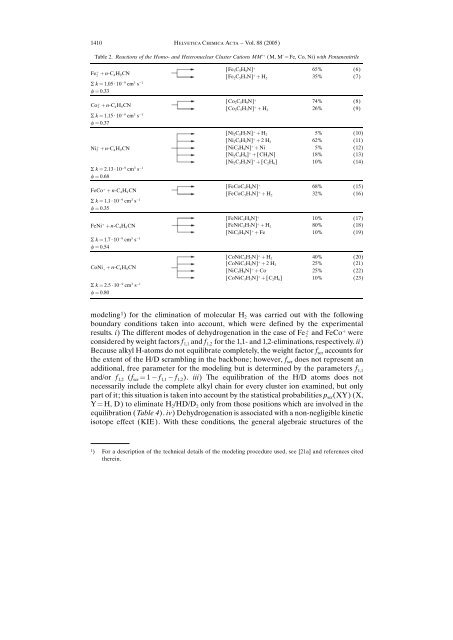
Table 2. <strong>Reactions</strong> <strong>of</strong> the <strong>Homo</strong>- <strong>and</strong> Heteronuclear Cluster Cations MM' ‡ (M, M' ˆ Fe, Co, Ni) with Pentanenitrile<br />
Fe ‡<br />
2 ‡ n-C4H9CN S k ˆ 1.05 ´ 10 9 cm3 s 1<br />
f ˆ 0.33<br />
[Fe 2C 5H 9N] ‡ 65% (6)<br />
[Fe 2C 5H 7N] ‡ ‡ H 2 35% (7)<br />
Co ‡ 2 ‡ n-C4H9CN [Co2C5H9N] ‡ [Co2C5H7N] 74% (8)<br />
‡ S k ˆ 1.15 ´ 10<br />
‡ H2 26% (9)<br />
9 cm3 s 1<br />
f ˆ 0.37<br />
[Ni2C5H7N] ‡ ‡ H2 5% (10)<br />
[Ni2C5H5N] ‡ ‡ 2H2 62% (11)<br />
Ni ‡ 2 ‡ n-C4H9CN [NiC5H9N] ‡ ‡ Ni 5% (12)<br />
[Ni2C4H6] ‡ ‡ [CH3N] 18% (13)<br />
[Ni2C3H3N] ‡ S k ˆ 2.13´ 10<br />
‡ [C2H6] 10% (14)<br />
9 cm3 s 1<br />
f ˆ 0.68<br />
FeCo ‡ ‡ n-C 4H 9CN<br />
S k ˆ 1.1 ´ 10 9 cm 3 s 1<br />
f ˆ 0.35<br />
[FeCoC 5H 9N] ‡ 68% (15)<br />
[FeCoC 5H 7N] ‡ ‡ H 2 32% (16)<br />
[FeNiC 5H 9N] ‡ 10% (17)<br />
FeNi ‡ ‡ n-C 4H 9CN [FeNiC 5H 7N] ‡ ‡ H 2 80% (18)<br />
[NiC 5H 9N] ‡ ‡ Fe 10% (19)<br />
S k ˆ 1.7 ´ 10 9 cm 3 s 1<br />
f ˆ 0.54<br />
CoNi ‡ ‡ n-C 4H 9CN<br />
S k ˆ 2.5 ´ 10 9 cm 3 s 1<br />
f ˆ 0.80<br />
[CoNiC5H7N] ‡ ‡ H2 40% (20)<br />
[CoNiC5H5N] ‡ ‡ 2H2 25% (21)<br />
[NiC5H9N] ‡ ‡ Co 25% (22)<br />
[CoNiC3H3N] ‡ ‡ [C2H6] 10% (23)<br />
modeling 1 ) for the elimination <strong>of</strong> molecular H 2 was carried out with the following<br />
boundary conditions taken into account, which were defined by the experimental<br />
results. i) The different modes <strong>of</strong> dehydrogenation in the case <strong>of</strong> Fe ‡ 2 <strong>and</strong> FeCo ‡ were<br />
considered by weight factors f 1,1 <strong>and</strong> f 1,2 for the 1,1- <strong>and</strong> 1,2-eliminations, respectively. ii)<br />
Because alkyl H-atoms do not equilibrate completely, the weight factor f scr accounts for<br />
the extent <strong>of</strong> the H/D scrambling in the backbone; however, f scr does not represent an<br />
additional, free parameter for the modeling but is determined by the parameters f 1,1<br />
<strong>and</strong>/or f 1,2 (f scr ˆ 1 f 1,1 f 1,2). iii) The equilibration <strong>of</strong> the H/D atoms does not<br />
necessarily include the complete alkyl chain for every cluster ion examined, but only<br />
part <strong>of</strong> it; this situation is taken into account by the statistical probabilities p scr(XY) (X,<br />
Yˆ H, D) to eliminate H 2/HD/D 2 only from those positions which are involved in the<br />
equilibration (Table 4).iv) Dehydrogenation is associated with a non-negligible kinetic<br />
isotope effect (KIE). With these conditions, the general algebraic structures <strong>of</strong> the<br />
1 ) For a description <strong>of</strong> the technical details <strong>of</strong> the modeling procedure used, see [21a] <strong>and</strong> references cited<br />
therein.




![Photoswitchable ionophores based on 1,3-alternate calix[4]arenes ...](https://img.yumpu.com/12290271/1/190x253/photoswitchable-ionophores-based-on-13-alternate-calix4arenes-.jpg?quality=85)