A Toy Model of Chemical Reaction Networks - TBI - Universität Wien
A Toy Model of Chemical Reaction Networks - TBI - Universität Wien
A Toy Model of Chemical Reaction Networks - TBI - Universität Wien
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
46 CHAPTER 5. REACTION NETWORKS<br />
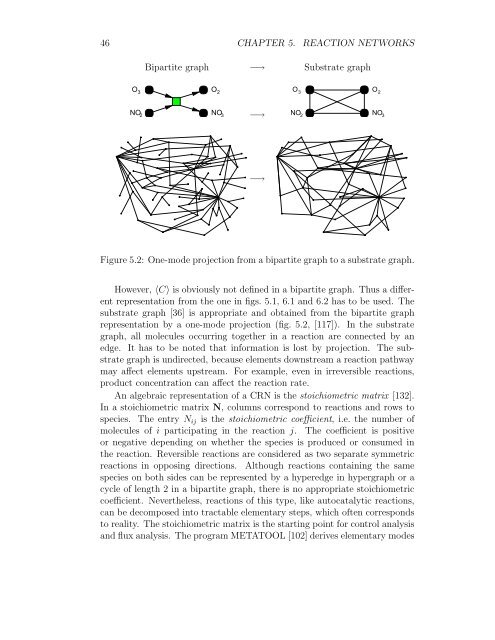
Bipartite graph −→ Substrate graph<br />
O 3<br />
NO 3<br />
NO 2<br />
O 2<br />
−→<br />
O 3<br />
NO 3<br />
NO 2<br />
O 2<br />
−→<br />
Figure 5.2: One-mode projection from a bipartite graph to a substrate graph.<br />
However, 〈C〉 is obviously not defined in a bipartite graph. Thus a different<br />
representation from the one in figs. 5.1, 6.1 and 6.2 has to be used. The<br />
substrate graph [36] is appropriate and obtained from the bipartite graph<br />
representation by a one-mode projection (fig. 5.2, [117]). In the substrate<br />
graph, all molecules occurring together in a reaction are connected by an<br />
edge. It has to be noted that information is lost by projection. The substrate<br />
graph is undirected, because elements downstream a reaction pathway<br />
may affect elements upstream. For example, even in irreversible reactions,<br />
product concentration can affect the reaction rate.<br />
An algebraic representation <strong>of</strong> a CRN is the stoichiometric matrix [132].<br />
In a stoichiometric matrix N, columns correspond to reactions and rows to<br />
species. The entry N ij is the stoichiometric coefficient, i.e. the number <strong>of</strong><br />
molecules <strong>of</strong> i participating in the reaction j. The coefficient is positive<br />
or negative depending on whether the species is produced or consumed in<br />
the reaction. Reversible reactions are considered as two separate symmetric<br />
reactions in opposing directions. Although reactions containing the same<br />
species on both sides can be represented by a hyperedge in hypergraph or a<br />
cycle <strong>of</strong> length 2 in a bipartite graph, there is no appropriate stoichiometric<br />
coefficient. Nevertheless, reactions <strong>of</strong> this type, like autocatalytic reactions,<br />
can be decomposed into tractable elementary steps, which <strong>of</strong>ten corresponds<br />
to reality. The stoichiometric matrix is the starting point for control analysis<br />
and flux analysis. The program METATOOL [102] derives elementary modes