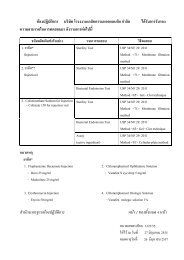
The Laboratory of Siam Bheasach Co., Ltd. has been accepted as ...
The Laboratory of Siam Bheasach Co., Ltd. has been accepted as ...
The Laboratory of Siam Bheasach Co., Ltd. has been accepted as ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
<strong>The</strong> <strong>Laboratory</strong> <strong>of</strong> <strong>Siam</strong> <strong>Bhe<strong>as</strong>ach</strong> <strong>Co</strong>., <strong>Ltd</strong>. <strong>h<strong>as</strong></strong> <strong>been</strong> <strong>accepted</strong> <strong>as</strong> an<br />
accredited laboratory in the field <strong>of</strong> drug testing for the following scopes.<br />
Type <strong>of</strong> Sample Test Method<br />
9. Sterile water for injection - pH<br />
(<strong>Co</strong>ntinued)<br />
- Volume in container<br />
- Particulate matter<br />
- Sterility USP 30 / NF 25 : 2007<br />
- Bacterial endotoxin<br />
- Ammonia<br />
10. Meropenem injection - Assay Meropenem<br />
(Mapenem 0.5 g injection,<br />
Mapenem 1.0 g injection)<br />
- Uniformity <strong>of</strong> dosage unit<br />
- pH<br />
- Loss on drying<br />
- Particulate matter<br />
USP 32 / NF 27 : 2009<br />
- <strong>Co</strong>ntent <strong>of</strong> sodium<br />
- Chromatographic purity<br />
- <strong>Co</strong>nstituted solution<br />
- Sterility test<br />
- Bacterial endotoxin<br />
- Identification<br />
Bureau <strong>of</strong> <strong>Laboratory</strong> Quality Standards Page 6 <strong>of</strong> 8<br />
Accreditation Number 1012/45<br />
Date <strong>of</strong> Accreditation : 20 January 2011<br />
Date <strong>of</strong> Expiration : 19 January 2014