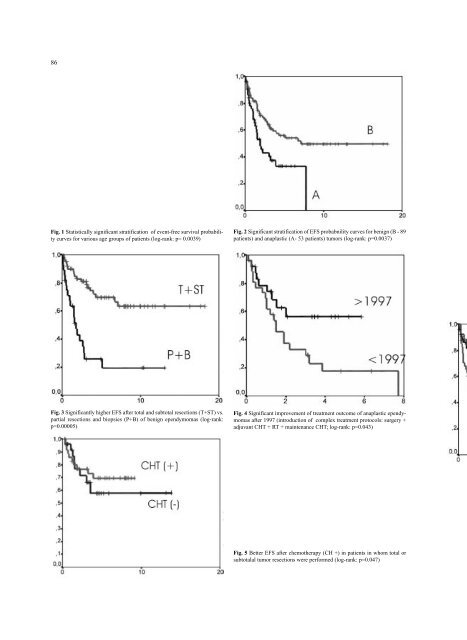
86 Fig. 1 Statistically significant stratification <strong>of</strong> event-free survival probability curves for various age groups <strong>of</strong> patients (log-rank: p= 0.0039) Fig. 2 Significant stratification <strong>of</strong> EFS probabnility curves for benign (B - 89 patients) and anaplastic (A- 53 patients) tumors (log-rank: p=0.0037) Fig. 3 Significantly higher EFS after total and subtotal resections (T+ST) vs. partial resections and biopsies (P+B) <strong>of</strong> benign ependymomas (log-rank: p=0.00005) Fig. 4 Significant improvement <strong>of</strong> treatment outcome <strong>of</strong> anaplastic ependymomas after 1997 (introduction <strong>of</strong> complex treatment protocols: surgery + adjuvant CHT + RT + maintenance CHT; log-rank: p=0.043) Fig. 5 Better EFS after chemotherapy (CH +) in patients in whom total or subtotalal tumor resections were performed (log-rank: p=0.047)
87 nancy in ependymomas [7, 21]. Opinions on adjuvant therapy differ from series to series. It is generally accepted that radiotherapy and chemotherapy can delay tumor recurrence but their influence on overall survival is still unclear [1, 5, 23, 28]. The rationale for agressive removal <strong>of</strong> ependymomas in certain locations is debatable. Devastating late complications <strong>of</strong> neuroaxis radiotherapy, especially in the youngest age group are well known [17, 18, 32]. That is why there is a trend to eliminate radiotherapy in children under 3 years <strong>of</strong> age and after total removal <strong>of</strong> histologically benign tumors. The fields <strong>of</strong> irradiation were restricted to tumor bed or tumor bed and ventricles for partially resected benign and anaplastic lesions respectively. The neuroaxis irradiation is usually reserved for disseminated ependymomas [12, 26]. This tendency is supported by the observations that reccurences are limited to primary tumor location in almost all cases and CSF dissemination is relatively rare [13]. Patterns <strong>of</strong> failure indicate, that final treatment outcome depends mostly on the local control <strong>of</strong> disease. These statements were the reason for the concept <strong>of</strong> “second-look” surgery, whenever reoperation was possible in cases <strong>of</strong> residual disease [8]. Chemotherapy was introduced in the treatment <strong>of</strong> children under 3 years to delay or eliminate the need for radiotherapy as well as in the treatment <strong>of</strong> malignant lesions and tumor recurrences and disseminations and finally in the treatment <strong>of</strong> partially resected benign lesions [15, 37]. Variable response rates to the same treatment protocols created the need for potential identification <strong>of</strong> subpopulations sensitive or resistent to chemotherapy, developement <strong>of</strong> novel drugs and intensification <strong>of</strong> treatment with subsequent bone marrow reconstruction [10, 14, 22]. As mentioned above, in our series neuroaxis irradiation correleated with better actuarial EFS for benign ependymomas. Significant change <strong>of</strong> treatment protocols over the years resulted in fairly confusing data. Before 1996, all cases were treated with neuroaxis irradiation; after 1996, RT was reserved only for disseminated ependymomas. It was clearly shown that neuroaxis RT did not improve EFS in anaplastic ependymomas in our series. An introduction <strong>of</strong> standarized treatment protocols (including chemotherapy) in 1997 improved treatment outcomes in malignant tumors. Chemotherapy also improved control <strong>of</strong> small tumor residues after surgery. Treatment response <strong>of</strong> tiny remnants seems to be an interesting clinical model for evaluation <strong>of</strong> CHT efficacy in the treatment <strong>of</strong> ependymomas. Conclusions 1. For the entire group <strong>of</strong> patients, age, extent <strong>of</strong> surgical resection, and histological malignancy, significantly influenced EFS; 2. Extent <strong>of</strong> surgical resection significantly influenced EFS in benign tumors and did not influence EFS in anaplastic ependymomas; 3. Neuroaxis RT did not influence EFS in patients with anaplastic ependymomas; 4. Treatment results in anaplastic ependymomas improved after 1997 (a potential role <strong>of</strong> CHT); 5. CHT significantly improved control <strong>of</strong> small tumor residues. References 1. Bouffet E, Foreman N (1999) Chemotherapy for intracranial ependymomas (review). Child’s Nerv Syst 15:563-323 2. Bouffet E, Perilongo G, Canete A, et al (1998) Intracranial ependymomas in children: a critical review <strong>of</strong> prognostic factors and a plea for cooperation. Medical and Pediatric Oncology 30:319-331 3. Duffner PK, Krischer JP, Sanford RA, et al (1998) Prognostic factors in infants and very young children with intracranial ependymomas. Pediatr Neurosurg 28:215-222 4. Ernestus R, Schroder R, Stutzer H, et al (1996) Prognostic relevance <strong>of</strong> localization and grading in intracranial ependymomas <strong>of</strong> childhood. Child’s Nerv Syst 12:522-526 5. Evans AE, Anderson JR, Lefkowitz- Boudreaux IB, et al (1996) Adjuvant chemotherapy <strong>of</strong> childhood posterior fossa ependymoma: caraniospinal irradiation with or without adjuvant CCNU, vincristine, and prednisone: a Childrens Cancer Group study. Med Pediatr Oncol 27:8-14 6. Figarella-Branger D, Civatte M, Bouvier-Labit C, et al (2000) Prognostic factors in intracranial ependymomas in children. J Neurosurg 93:605-613 7. Figarella-Branger D, Gambarelli D, Dollo C, et al (1991) Infratentorial ependymomas <strong>of</strong> childhood. Correlation between histological features, immunohistological fenotype, silver nucleolar staining values and post-operative survival in 16 cases; Acta Neuropathol 82:208-216 8. Foreman NK, Love S, Gill SS, Coakham HB (1997) Second-look surgery for incompletely resected fourth ventricle ependymomas: technical case report. Neurosurgery 40:856-860 9. Foreman NK, Love S, Thorne R (1996) Intracranial ependymomas: analysis <strong>of</strong> prognostic factors in a population-based series. Pediatr Neurosurg 24:119-125 10. Geddes JF, Vowles GH, Ashmore SM (1994) Detection <strong>of</strong> multidrug resistance gene product (P-glycoprotein) expression in ependymomas. Neuropathol Appl Neurobiol 20(2):118-121 11. Gerszten PC, Pollack IF, Martinez AJ (1996) Intracranial ependymomas <strong>of</strong> childhood. Lack <strong>of</strong> correlation <strong>of</strong> histopathology and clinical outcome. Path Res Pract 192:515-522 12. Goldwein JW, Corn BW, Finlay JL, et al (1991) Is craniospinal irradiation required to cure children with malignant (anaplastic) intractranial ependymomas Cancer 67:2766-2771 13. Goldwein JW, Glause TA, Packer RJ, et al (1990) Recurrent intracranial ependymomas in children. Survival, patterns <strong>of</strong> feilure, and prognostic factors. Cancer 66: 557-563 14. Grill J, Kalifa C, Doz F, et al (1996) A high-dose busulfan thiotepa combination followed by autologous bone marrow transplantation in childhood recurrent ependymoma. A phase II study. Pediatr Neurosurg 25:7-12 15. Grill J, LeDeley MC, Gambarelli D, et al (2001) Postoperative chemotherapy without irradiation for ependymoma in children under 5 years <strong>of</strong> age: a multicenter trial <strong>of</strong> the French Society <strong>of</strong> Pediatric Oncology. J Clin Oncol 19:1288-1296