Suren Kandasamy Dissertation.pdf - University of Surrey
Suren Kandasamy Dissertation.pdf - University of Surrey
Suren Kandasamy Dissertation.pdf - University of Surrey
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
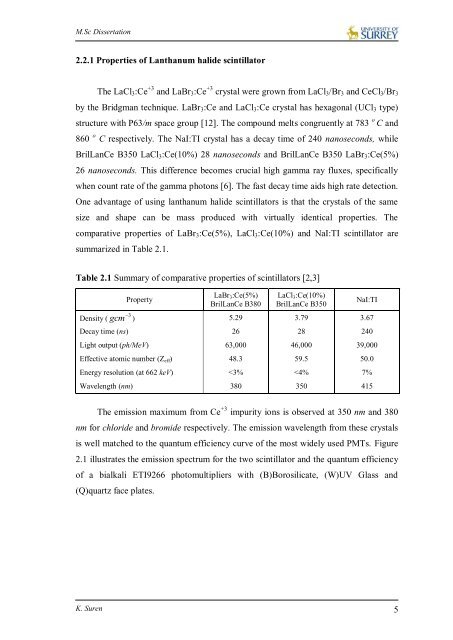
M.Sc <strong>Dissertation</strong>2.2.1 Properties <strong>of</strong> Lanthanum halide scintillatorThe LaCl 3 :Ce +3 and LaBr 3 :Ce +3 crystal were grown from LaCl 3 /Br 3 and CeCl 3 /Br 3by the Bridgman technique. LaBr 3 :Ce and LaCl 3 :Ce crystal has hexagonal (UCl 3 type)structure with P63/m space group [12]. The compound melts congruently at 783 o C and860 o C respectively. The NaI:TI crystal has a decay time <strong>of</strong> 240 nanoseconds, whileBrilLanCe B350 LaCl 3 :Ce(10%) 28 nanoseconds and BrilLanCe B350 LaBr 3 :Ce(5%)26 nanoseconds. This difference becomes crucial high gamma ray fluxes, specificallywhen count rate <strong>of</strong> the gamma photons [6]. The fast decay time aids high rate detection.One advantage <strong>of</strong> using lanthanum halide scintillators is that the crystals <strong>of</strong> the samesize and shape can be mass produced with virtually identical properties. Thecomparative properties <strong>of</strong> LaBr 3 :Ce(5%), LaCl 3 :Ce(10%) and NaI:TI scintillator aresummarized in Table 2.1.Table 2.1 Summary <strong>of</strong> comparative properties <strong>of</strong> scintillators [2,3]Density (PropertyLaBr 3 :Ce(5%)BrilLanCe B380LaCl 3 :Ce(10%)BrilLanCe B350NaI:TI3gcm ) 5.29 3.79 3.67Decay time (ns) 26 28 240Light output (ph/MeV) 63,000 46,000 39,000Effective atomic number (Z eff ) 48.3 59.5 50.0Energy resolution (at 662 keV)