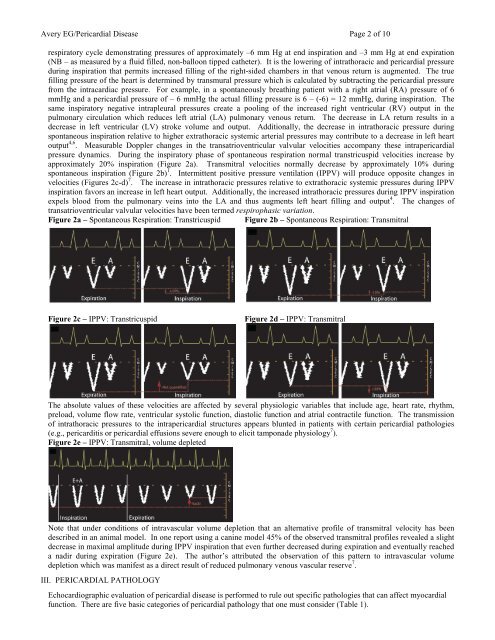
Avery EG/<strong>Pericardial</strong> <strong>Disease</strong> Page 2 <strong>of</strong> 10respiratory cycle demonstrating pressures <strong>of</strong> approximately –6 mm Hg at end inspiration and –3 mm Hg at end expiration(NB – as measured by a fluid filled, non-balloon tipped catheter). It is the lowering <strong>of</strong> intrathoracic and pericardial pressureduring inspiration that permits increased filling <strong>of</strong> the right-sided chambers in that venous return is augmented. The truefilling pressure <strong>of</strong> the heart is determined by transmural pressure which is calculated by subtracting the pericardial pressurefrom the intracardiac pressure. For example, in a spontaneously breathing patient with a right atrial (RA) pressure <strong>of</strong> 6mmHg and a pericardial pressure <strong>of</strong> – 6 mmHg the actual filling pressure is 6 – (-6) = 12 mmHg, during inspiration. Thesame inspiratory negative intrapleural pressures create a pooling <strong>of</strong> the increased right ventricular (RV) output in thepulmonary circulation which reduces left atrial (LA) pulmonary venous return. The decrease in LA return results in adecrease in left ventricular (LV) stroke volume and output. Additionally, the decrease in intrathoracic pressure duringspontaneous inspiration relative to higher extrathoracic systemic arterial pressures may contribute to a decrease in left heartoutput 4,6 . Measurable Doppler changes in the transatrioventricular valvular velocities ac<strong>com</strong>pany these intrapericardialpressure dynamics. During the inspiratory phase <strong>of</strong> spontaneous respiration normal transtricuspid velocities increase byapproximately 20% inspiration (Figure 2a). Transmitral velocities normally decrease by approximately 10% duringspontaneous inspiration (Figure 2b) 1 . Intermittent positive pressure ventilation (IPPV) will produce opposite changes invelocities (Figures 2c-d) 7 . The increase in intrathoracic pressures relative to extrathoracic systemic pressures during IPPVinspiration favors an increase in left heart output. Additionally, the increased intrathoracic pressures during IPPV inspirationexpels blood from the pulmonary veins into the LA and thus augments left heart filling and output 4 . The changes <strong>of</strong>transatrioventricular valvular velocities have been termed respirophasic variation.Figure 2a – Spontaneous Respiration: Transtricuspid Figure 2b – Spontaneous Respiration: TransmitralFigure 2c – IPPV: TranstricuspidFigure 2d – IPPV: TransmitralThe absolute values <strong>of</strong> these velocities are affected by several physiologic variables that include age, heart rate, rhythm,preload, volume flow rate, ventricular systolic function, diastolic function and atrial contractile function. The transmission<strong>of</strong> intrathoracic pressures to the intrapericardial structures appears blunted in patients with certain pericardial pathologies(e.g., pericarditis or pericardial effusions severe enough to elicit tamponade physiology 7 ).Figure 2e – IPPV: Transmitral, volume depletedNote that under conditions <strong>of</strong> intravascular volume depletion that an alternative pr<strong>of</strong>ile <strong>of</strong> transmitral velocity has beendescribed in an animal model. In one report using a canine model 45% <strong>of</strong> the observed transmitral pr<strong>of</strong>iles revealed a slightdecrease in maximal amplitude during IPPV inspiration that even further decreased during expiration and eventually reacheda nadir during expiration (Figure 2e). The author’s attributed the observation <strong>of</strong> this pattern to intravascular volumedepletion which was manifest as a direct result <strong>of</strong> reduced pulmonary venous vascular reserve 7 .III. PERICARDIAL PATHOLOGY<strong>Echocardiographic</strong> evaluation <strong>of</strong> pericardial disease is performed to rule out specific pathologies that can affect myocardialfunction. There are five basic categories <strong>of</strong> pericardial pathology that one must consider (Table 1).
Avery EG/<strong>Pericardial</strong> <strong>Disease</strong> Page 3 <strong>of</strong> 10Table 1. General Categories <strong>of</strong> <strong>Pericardial</strong> PathologyCongenital <strong>Pericardial</strong> DefectsPericarditis<strong>Pericardial</strong> Effusion<strong>Pericardial</strong> Tamponade<strong>Pericardial</strong> MassesCongenital pericardial defects are rare and consist <strong>of</strong> partial or total absence <strong>of</strong> the pericardium and Mulibrey nanism, anautosomal recessive disorder, primarily found in the Finnish population that can lead to congestive heart failure in early adultlife and is partially characterized by the development <strong>of</strong> constrictive pericarditis in some affected individuals. TEE findings <strong>of</strong>constrictive pericarditis will be discussed in a later section. The significance <strong>of</strong> pericardial absence varies although appearsminor in that most <strong>of</strong> these patients are clinically asymptomatic (Table 2). One small observational study (n=10) reviewingthis disorder found that paroxysmal stabbing chest pain that mimicked coronary ischemia was the most <strong>com</strong>mon clinicalpresentation 8 . Individuals with congenital absence <strong>of</strong> the pericardium are at increased risk for additional congenitalabnormalities in that 30% <strong>of</strong> these patients will have some additional congenital pathology 3,23,24 .Table 2. Pathologic Characteristics <strong>of</strong> Forms <strong>of</strong> <strong>Pericardial</strong> AbsenceFormIncidence Clinical SignificanceTotal Bilateral Absence Rare Mostly asymptomaticPartial Left Absence 70% Increased risk for aortic dissection if augmented heart mobility;possible herniation or strangulation <strong>of</strong> heart structures throughthe defect with chest pain, shortness <strong>of</strong> breath, syncopePartial Right Absence 17% Increased risk for aortic dissection if augmented mobility<strong>of</strong> the heart exists as a result <strong>of</strong> the defectOverall 0.01%Incidence values represent a percentage <strong>of</strong> the overall incidence <strong>of</strong> 0.01%.<strong>Echocardiographic</strong>ally, one may observe paradoxic ventricular septal motion, an exaggerated posterior left ventricular wall andpossibly the appearance <strong>of</strong> a dilated right ventricle when assessed through the transthoracic parasternal window. A small series<strong>of</strong> patients (n=8) from a Japanese group reported more marked alterations in venous return in the right heart (measured in thesuperior vena cava) than in the left heart (measured in the pulmonary veins) 9 . CXR and cardiac MRI are the two imagingmodalities that appear to be most useful in making the definitive diagnosis <strong>of</strong> congenital absence <strong>of</strong> the pericardium 9 .Pericarditis is an inflammation <strong>of</strong> the pericardium and can occur as the result <strong>of</strong> multiple pathologies. Pericarditis maypresent as either a chronic or acute process and is <strong>of</strong>ten associated with the development <strong>of</strong> a pericardial effusion. Clinically,one examines the patient suspected <strong>of</strong> having pericarditis for the presence <strong>of</strong> the classic triad <strong>of</strong> chest pain, ECG evidence <strong>of</strong>pericarditis (e.g., diffuse ST segment elevations), and a pericardial rub on auscultation. Etiologies <strong>of</strong> pericarditis include thefollowing:• Infections (e.g., postviral, bacterial-especially tuberculosis)• Neoplastic (e.g., primary-mesothelioma or fibrosar<strong>com</strong>a; secondary metastatic disease-melanoma, lymphoma, leukemia or directextension <strong>of</strong> a pulmonic or breast tumor)• Immune/Inflammatory (e.g., rheumatoid arthritis, systemic lupus erythematosus, scleroderma, acute rheumatic fever, dermatomyositis,Wegener’s granulomatosis, mixed collagen vascular disease; post-myocardial infarction (Dressler’s syndrome); uremia; postcardiacsurgery• Intracardiac-pericardial <strong>com</strong>munications (e.g., chest trauma, post-interventional catheter procedures, postmyocardial ventricularrupture) 2 .<strong>Echocardiographic</strong>ally, one inspects the pericardium for evidence <strong>of</strong> thickening that appears as an increase in the brightness, orechogenicity <strong>of</strong> the ultrasound signal. Normal pericardial thickness is approximately 2-3 mm. M-mode analysis <strong>of</strong> a patientwith pericarditis will reveal multiple parallel ultrasound reflections and can be helpful to discern the thickened pericardium(Figure 3). Multiple echo windows should be obtained to verify the diffuse or localized nature <strong>of</strong> the pericarditis.Echocardiography lacks sufficient fidelity to consistently quantify the degree <strong>of</strong> pericarditis and thus carries an unreliablesensitivity and specificity for detecting this disorder <strong>com</strong>pared to other imaging techniques (e.g, cardiac MRI or high resolution(64 slice) <strong>com</strong>puted tomography).