AD 2016 Q1
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
RESEARCH, EDUCATION & MEDICINE<br />
EXPERT OPINIONS<br />
At each of these three stages there are factors that<br />
modify the effects and outcomes of exposure. In the<br />
absence of specific knowledge, the most common<br />
preventive strategy is to reduce supersaturation by<br />
limiting the depth/time exposure and ascent rate for<br />
all divers to avoid substantial bubbling — even though<br />
bubbles put only some divers (those with a right-toleft<br />
shunt [RLS]) at risk. Without bubbles (or with<br />
only a few), there will be no arterialization, even in<br />
divers with RLS, and there will be no DCS in those<br />
susceptible to arterial embolization.<br />
Several approaches to mitigating postdive bubble<br />
occurrence have been studied; these include predive<br />
removal of hypothetical bubble nuclei by wholebody<br />
vibration, attempts to influence oxygen radicals<br />
or nitric oxide suspected of contributing to bubble<br />
generation, stimulation of heat-shock protein<br />
production and various other methods, including<br />
predive chocolate treats. Although some of these<br />
factors may reduce the amount of bubbles, the effects<br />
vary and may be of less importance than the individual<br />
variation in response to decompression.<br />
If every diver had a consistent individual propensity<br />
for VGE production and could be classified as a<br />
“bubbler” or “nonbubbler” across a broad spectrum of<br />
reasonable dive exposures, safe exposure limits could be<br />
tailored individually. We would achieve greater precision<br />
if we could identify those divers who have persistent<br />
or occasional RLS and then customize a dive exposure<br />
for them. At present we already know that a large<br />
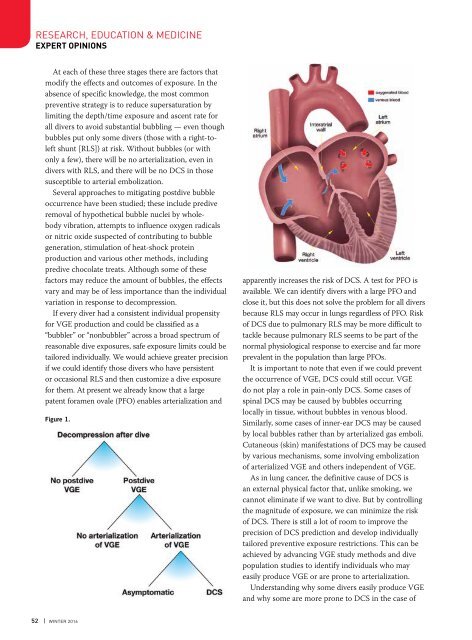
patent foramen ovale (PFO) enables arterialization and<br />
Figure 1.<br />
apparently increases the risk of DCS. A test for PFO is<br />
available. We can identify divers with a large PFO and<br />
close it, but this does not solve the problem for all divers<br />
because RLS may occur in lungs regardless of PFO. Risk<br />
of DCS due to pulmonary RLS may be more difficult to<br />
tackle because pulmonary RLS seems to be part of the<br />
normal physiological response to exercise and far more<br />
prevalent in the population than large PFOs.<br />
It is important to note that even if we could prevent<br />
the occurrence of VGE, DCS could still occur. VGE<br />
do not play a role in pain-only DCS. Some cases of<br />
spinal DCS may be caused by bubbles occurring<br />
locally in tissue, without bubbles in venous blood.<br />
Similarly, some cases of inner-ear DCS may be caused<br />
by local bubbles rather than by arterialized gas emboli.<br />
Cutaneous (skin) manifestations of DCS may be caused<br />
by various mechanisms, some involving embolization<br />
of arterialized VGE and others independent of VGE.<br />
As in lung cancer, the definitive cause of DCS is<br />
an external physical factor that, unlike smoking, we<br />
cannot eliminate if we want to dive. But by controlling<br />
the magnitude of exposure, we can minimize the risk<br />
of DCS. There is still a lot of room to improve the<br />
precision of DCS prediction and develop individually<br />
tailored preventive exposure restrictions. This can be<br />
achieved by advancing VGE study methods and dive<br />
population studies to identify individuals who may<br />
easily produce VGE or are prone to arterialization.<br />
Understanding why some divers easily produce VGE<br />
and why some are more prone to DCS in the case of<br />
52 | WINTER <strong>2016</strong>