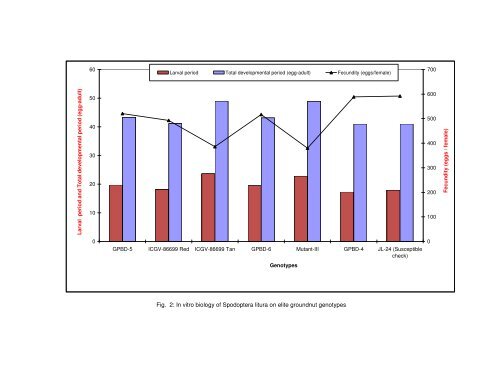
ecome an <strong>in</strong>tegral part <strong>of</strong> the process <strong>of</strong> identify<strong>in</strong>g <strong>and</strong> study<strong>in</strong>g pest resistance <strong>in</strong> <strong>groundnut</strong> (Prasad et al., 2000). Groundnut <strong>genotypes</strong> such as ICGV- 86699 Tan, Mutant III, GPBD-6, GPBD-5, ICGV- 86699 Red, GPBD-4 <strong>and</strong> susceptible check (JL-24), were utilized for rear<strong>in</strong>g S. litura. The effect <strong>of</strong> these <strong>genotypes</strong> on larval, pre pupal, pupal, pre oviposition, oviposition <strong>and</strong> post oviposition period, larval weight, larval mortality, per cent pupal survival ,pupal weight ,adult longevity, per cent adult emergence <strong>and</strong> fecundity <strong>of</strong> S. litura was ascerta<strong>in</strong>ed on each <strong>of</strong> the <strong>elite</strong> <strong>genotypes</strong>. The present study demonstrated existence <strong>of</strong> substantial amount <strong>of</strong> variability <strong>in</strong> host, affect<strong>in</strong>g these biological parameters. 5.1.1.1 Larval period The length <strong>of</strong> larval duration was affected <strong>in</strong> larvae fed on foliage <strong>of</strong> test <strong>genotypes</strong> (Table 2) <strong>and</strong> there by reduces biotic potential <strong>of</strong> the pest. The <strong>genotypes</strong> Mutant III <strong>and</strong> ICGV- 86699 Tan recorded longer larval duration <strong>in</strong> each <strong>in</strong>stars compare to other <strong>genotypes</strong>. While <strong>in</strong> susceptible <strong>genotypes</strong> viz., GPBD-4 <strong>and</strong> JL-24 recorded shorter larval duration (Fig. 3). The present study corroborate with the f<strong>in</strong>d<strong>in</strong>gs <strong>of</strong> Patil et al. (1995) where <strong>in</strong> S. litura had stretched larval duration on ICGV-87165, ICGV- 86350 <strong>and</strong> ICGV- 87264. Bioassay carried out with the larvae to underst<strong>and</strong> the mechanism <strong>of</strong> resistance by Wightman <strong>and</strong> Ranga Rao (1994) revealed no antibiosis effect on II <strong>and</strong> IV <strong>in</strong>star larvae when fed to matured leaves <strong>of</strong> ICGV- 86031. Spodoptera frugiperda (S.) fed with resistant florunner took more days to develop compared to larvae fed with curly leaf (Todd et al., 1991). It has also been showed that longer larval duration on resistant <strong>genotypes</strong>, NC Ac -2243 (Xi Jia LI , 1987) was longer. 5.1.1.2 Larval weight <strong>and</strong> larval mortality The <strong>genotypes</strong> Mutant III <strong>and</strong> ICGV- 86699 Tan recorded significantly low larval weight <strong>and</strong> high percentage <strong>of</strong> mortality at all the stages compared to susceptible <strong>genotypes</strong> GPBD-4 <strong>and</strong> JL-24 (Table 3 <strong>and</strong> Fig. 2). The larval per cent mortality was high on resistant <strong>genotypes</strong> <strong>in</strong> early stages compare to susceptible <strong>genotypes</strong> <strong>in</strong>dicat<strong>in</strong>g the vulnerability <strong>of</strong> neonate larvae to the exist<strong>in</strong>g resistant factor. Accord<strong>in</strong>g to Stevenson et al. (1993) <strong>in</strong> pest control strategies, neonate larvae should be a primary target <strong>in</strong> host plant resistance because plant damage can be m<strong>in</strong>imized if pest is elim<strong>in</strong>ated as early <strong>in</strong> the life cycle as possible. The higher larval mortality <strong>of</strong> S.litura on resistant <strong>groundnut</strong> <strong>genotypes</strong> like ICGV-86031, wild tetraploid Arachis manticola was also reported by many workers (Kulkarni, 1989; Dwivedi et al., 1993; Wightman <strong>and</strong> Ranga Rao, 1994; Patil et al., 1995; Prasad <strong>and</strong> Gowda, 2006). Mortality at early stages has also been observed <strong>in</strong> Heliothis zea when reared on maize plant. The development <strong>of</strong> first stadium larvae <strong>of</strong> H. zea was retarded by the presence <strong>of</strong> chlorogenic acid <strong>and</strong> rut<strong>in</strong> <strong>in</strong> artificial diet (Isman <strong>and</strong> Duffey, 1982). Present f<strong>in</strong>d<strong>in</strong>gs corroborates with the f<strong>in</strong>d<strong>in</strong>gs <strong>of</strong> Prasad <strong>and</strong> Gowda (2006) where <strong>in</strong> the larval weight was significantly low from larvae fed on the foliage <strong>of</strong> resistant <strong>genotypes</strong> NC Ac 343, Mutant 28-2 <strong>and</strong> R 9227. S<strong>in</strong>gh <strong>and</strong> Sachan (1992) identified ICGV-86030, ICGV- 86031 <strong>and</strong> NC Ac 343 as resistant to S. litura based on survival, weight ga<strong>in</strong> <strong>and</strong> larval duration. The differential response <strong>of</strong> the <strong>genotypes</strong> on larval parameters <strong>in</strong>dicates the possibility <strong>of</strong> antibiosis mechanisms <strong>of</strong> resistance operat<strong>in</strong>g <strong>in</strong> them. The effect <strong>of</strong> resistant <strong>genotypes</strong> on larval mortality <strong>in</strong> early stage, ga<strong>in</strong> <strong>in</strong> larval weight <strong>and</strong> growth <strong>of</strong> the larvae could obviously be due to chemical factors, i.e. antibiosis as elucidated by Pa<strong>in</strong>ter (1951). The chemicals viz., querecit<strong>in</strong> glycosiden, chlorogenic acid <strong>and</strong> rut<strong>in</strong> have been reported to be the cause for resistance <strong>in</strong> wild Arachis species (Stevenson, 1993) <strong>and</strong> could be the cause for antibiosis. However, physical resistance (leaf thickness) may be also important as panitrometric studies showed that leaves <strong>of</strong> resistant wild Arachis species required a greater bit<strong>in</strong>g effort than did the leaves <strong>of</strong> susceptible TMV-2 <strong>and</strong> more susceptible <strong>of</strong> Arachis. 5.1.1.3 Pupal development <strong>and</strong> moth emergence The resistant effect <strong>of</strong> Mutant III <strong>and</strong> ICGV- 86699 Tan were also observed on pupal duration, pupal weight, per cent pupal survival <strong>and</strong> moth emergence (Table 5 <strong>and</strong> 4). Similar
Larval period <strong>and</strong> Total developmental period (egg-adult) 60 50 40 30 20 10 0 Larval period Total developmental period (egg-adult) Fecundity (eggs/female) GPBD-5 ICGV-86699 Red ICGV-86699 Tan GPBD-6 Mutant-III GPBD-4 JL-24 (Susceptible check) Genotypes Fig. 2: In vitro biology <strong>of</strong> Spodoptera litura on <strong>elite</strong> <strong>groundnut</strong> <strong>genotypes</strong> 700 600 500 400 300 200 100 0 Fecundity (eggs / female)
- Page 1 and 2:
SCREENING ELITE GENOTYPES AND IPM O
- Page 3 and 4: CONTENTS Sl. No. Chapter Particular
- Page 5 and 6: Figure No. LIST OF FIGURES Title 1.
- Page 7 and 8: In some parts of the northern Karna
- Page 9 and 10: mutants were systematically screene
- Page 11 and 12: observed on Dwarf Mutant, the lowes
- Page 13 and 14: Singh and Sachan (1991) recorded se
- Page 15 and 16: Thontadarya and Nangia (1983) repor
- Page 17 and 18: Monitoring with pheromone traps to
- Page 19 and 20: 1. JL-24 (Susceptible check) 2. ICG
- Page 21 and 22: Observations were recorded daily on
- Page 23 and 24: Table 1: Performance of elite groun
- Page 25 and 26: Third instar duration was significa
- Page 27 and 28: Table 3: Gain in larval weight and
- Page 29 and 30: Table 4: Biological parameters of S
- Page 31 and 32: Table 5: In vitro biology of Spodop
- Page 33 and 34: sunflower and N. rileyi, foxtail mi
- Page 35 and 36: Table 7: Leaf hoppers population in
- Page 37 and 38: Table 8: Thysanoplusia orichalcea p
- Page 39 and 40: Table 10: Spilarctia obliqua popula
- Page 41 and 42: 4.2.4.2 50 days after sowing The de
- Page 43 and 44: Table 13: The management of Spodopt
- Page 45 and 46: The early instar larvae scraped the
- Page 47 and 48: Module I Table 16: Natural enemy po
- Page 49 and 50: Table 18: Natural enemy population
- Page 51 and 52: Table 19: Economics of IPM modules
- Page 53: Per cent defoliation 50 45 40 35 30
- Page 57 and 58: Larval Larval weight weight (g) (g)
- Page 59 and 60: 5.2.3 Defoliator population in thre
- Page 61 and 62: Pest population 1600 1400 1200 1000
- Page 63 and 64: Gross returns and Net returns (Rs./
- Page 65 and 66: REFERENCES Agasimani, C. A., Ravish
- Page 67 and 68: Kennedy, F. J. S., Rajamanickam. K.
- Page 69 and 70: Prasad, M. N. R. and Gowda, M. V. C
- Page 71 and 72: Tiwari, S. N., Rathore, Y. S. and B
- Page 73: SCREENING ELITE GENOTYPES AND IPM O