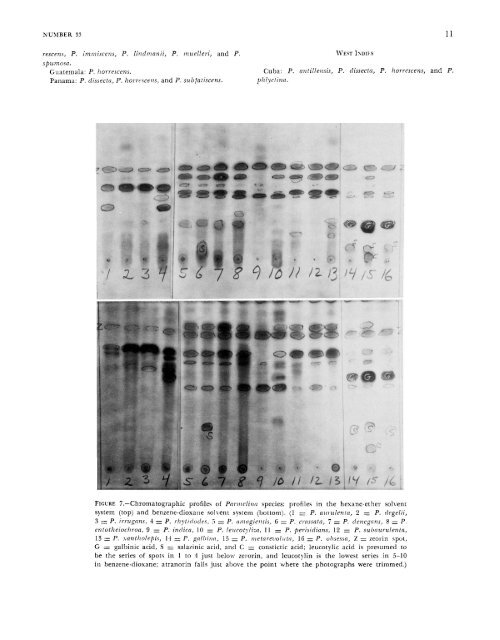
10 S\IITHSONIAS CONTRIBUTIONS TO BOTANY acid in P. metarevoluta. All <strong>of</strong> <strong>the</strong>se species, ex- cepting P. versiformis, are distributed in <strong>the</strong> Old World following a pattern seen in Bulbothrix (<strong>Hale</strong>, 1976b). Stictic acid has been discovered in only one spe- cies, P. mnelleri, where it is accompanied by con- stictic acid and two unknown compounds falling between stictic and constictic acids in benezene- dioxane. TERPENEL-A closely related series <strong>of</strong> triterpenes with a liopane skeleton have been described for species in section Myelochroa. Their structures have been determined by Yosioka and his group in Ja- pan using nuclear magnetic resonance (NhIR) spectra, mass spectrometry, etc. The tliin-layer chromatography is not yet standardized or well un- derstood, and I have not tried to identify individual spots. Typical pr<strong>of</strong>iles for <strong>the</strong> terpene-containing species <strong>of</strong> <strong>Parmelina</strong> are illustrated in Figure 7. Leucotylic acid, a colorless compound closely related to both zeorin and leucotylin, was first elu- cidated by Yosioka (1966). It was isolated from Pa?.- melina leucotyliza, although my results indicate that leucotylin is, in fact, <strong>the</strong> main component <strong>of</strong> this species. I suspect that <strong>the</strong>ir material included a mixture <strong>of</strong> P. crassata, which does contain leuco- tylic acid. The species <strong>of</strong> Pnrmelina which contain leucotylic acid (or at least have <strong>the</strong> same terpenic pr<strong>of</strong>ile as P. airrulentn, as illustrated in Figure 7) are P. aurzilenta, P. degelii, P. il-rugans, and P. rhytidodes. Leucotylin, <strong>the</strong> progenitor <strong>of</strong> leucotylic acid, is <strong>the</strong> main terpene component in P. amagiensis, P. crassata, P. denegans, P. ento<strong>the</strong>iochroa, P. galbina, P. hayachinensis, P. indica, P. leucotyliza, P. meta- revoluta, P. obsessa, P. perisidians, P. subaurulenta, and P. xantholepis. It forms a low major spot in benzene-dioxane, but I have generally identified it as a pr<strong>of</strong>ile <strong>of</strong> several terpene spots as illustrated in Figure 7. Yosioka (Yosioka and Nakanislii, 1963; Yosioka and Nakanishi, 1966) has isolated at least five o<strong>the</strong>r compounds related to leucotylin in “Parmelia ento- <strong>the</strong>iochroa.” I presume that at least some <strong>of</strong> <strong>the</strong>se make up <strong>the</strong> numerous spots resolved in hexane- e<strong>the</strong>r (see Figure 7), but no attempt has been made to identify any <strong>of</strong> <strong>the</strong>m on <strong>the</strong> plates. There seems to be great variation in <strong>the</strong> intensity <strong>of</strong> <strong>the</strong> spots, reflecting different concentrations in <strong>the</strong> thallus. It remains to be seen whe<strong>the</strong>r this variation has any taxonomic value. Zeorin is a well known lichen substance. The stereochemistry <strong>of</strong> its hopane skeleton was only recently studied by Yosioka et al. (1968b). It occurs in all species in section 1\4pelochroa and has been reported in Acroscyplirrs, Cladonin spp., Hppotrachpa majoris (Vainio) <strong>Hale</strong>, Lecanom, Sephroma, Peliigera, and <strong>the</strong> Physciaceae. It may be identified as <strong>the</strong> highest blue spot on <strong>the</strong> chromatographic plates in Iiotli solvent systems (Figure 7). Prc~iE.\’Ts.-.\Iedullary pigments are especially characteristic <strong>of</strong> species in section hfyelochroa. The chromatography <strong>of</strong> <strong>the</strong>se is extremely difficult and ~is~iallp unsuccessful. There is considerable streaking and individual components cannot be distinguished. Secalonic acid X was first ideniifietl by Yosioka et al. (1968a:2090), who established its identity with ento<strong>the</strong>in. It also occurs in ergot (Shibata et al., 1964). Yosioka found secalonic acid A in <strong>Parmelina</strong> nii~rilcntm, P. enfo<strong>the</strong>iochroa, P. pel-isiclians, and P. subauixlenta, and I presume it is <strong>the</strong> major pigment in all species <strong>of</strong> section ilfpelochroa with a yellowish orange medulla, as well as in P. immiscens and P. lintlmanii, both lacking terpenes. Unidentified reddish pigments accompany secalonic acid 4 in <strong>the</strong> lower medullary layer <strong>of</strong> P. amagien- ,pis and P. dcnegans. Phytogeography and Speciation Pawnelinti occurs primarily on trees in secondary forests in lemperate zones and at higher elevations in <strong>the</strong> tropics. A number <strong>of</strong> <strong>the</strong> commoner species may also occur on rocks. There are, however, only four oliligately saxicolous species, P. enownis, P. indica, P. obsessa, and P. usambaiensi.r. The Pnr- melina floras <strong>of</strong> various geopolitical units are enumerated below. It is altoge<strong>the</strong>r obvious that many countries are underrepresented because <strong>the</strong>y have not been visited by lichen collectors. XORTH AMERIC.~ United States: P. antillensis, P. aurulenta, P. dissecta, P. galbi17n, P. hoi 1 escciis, P. ~ne/ar~~volii/a, P. obsessa, and P. sfiu in osa. MEXICO ALD C~NTRAL AMERIC~ Mexico: P. antillensis, P. aurulenta, P. dissecta, P. hor-
NUMBER 33 11 rescens, P. ivztniscens, P. Zindnlanii, P. niuelieri, and P. WEIT ISDIE s spumosa. Guatemala: P. horrescens. Cuba: P. antiliensis, P. dissecta, P. horrescens, and P. Panama: P. dissect(/, P. Izorre,ce,ls, and P. subfutiscens. pizlyctinn. FIGURE 7.-Chromatographic pr<strong>of</strong>iles <strong>of</strong> Pnrnielinu species: pr<strong>of</strong>iles in <strong>the</strong> hexane-e<strong>the</strong>r solvent system (top) and benzene-dioxaiie solvent system (bottom). (1 1 P. uicndenla, 2 = P. degelii, 3 = P. irrugans, 4 = P. rhytidot/u.i, 5 = P. ci)nrrgie!i,iis, G = P. ci-nssata, 7 = P. denegnns, 8 = P. ento<strong>the</strong>iochroa, 9 = P. indlica, 10 = P. iettcotyliza, 11 = P. perisidians, 12 = P. subaurulentn, 13 = P. santholepis, 14 = P. plbi!rcr, 1.5 = P. tnetarevoltrtrr, lG = P. ohsessa, Z = zeorin spot, G = galbinic acid, S = salarinic acid, and C = constictic acid: leucotylic acid is presumed to be <strong>the</strong> series <strong>of</strong> spots in 1 to 4 just below rerorin, and leucotylin is <strong>the</strong> lowest series in 5-10 in benzene-dioxane; atranorin falls just above <strong>the</strong> point where <strong>the</strong> photographs were trimmed.)