20 SMITHSONIAN CONTRIBUTIONS TO BOTANY FIGURE 10.-Distribution <strong>of</strong> Parmefina aurzrlenta hased on all available herharium specimens. geographic areas but it does not seem significant or consistent. I did not, incidentally, recheck <strong>the</strong> terpene pr<strong>of</strong>iles <strong>of</strong> Pawnelia tiliacea var. efiol-esrens, P. albido-strarninea, P. subrevoluta, or P. hunanen- sis, but assumed that <strong>the</strong>y have <strong>the</strong> “aurulenta” ter- penes since <strong>the</strong>y are sorediate and fall outside <strong>the</strong> range <strong>of</strong> P. leucotyliza. Should any <strong>of</strong> <strong>the</strong>m be found to contain “subaurulenta” terpenes, <strong>the</strong> no- menclature <strong>of</strong> P. aurulenta would not change. The parent morph <strong>of</strong> Pal-melina awulenta might well be P. irl-ugans, which is identical in chemistry. It would be desirable, however, to examine more specimens <strong>of</strong> P. irrugans in relation to P. aurulenta in Japan before making a final decision. The ecology <strong>of</strong> P. aurulenta has been intensively studied in Wisconsin, an area <strong>of</strong> typical deciduous and conifer forests well within its range. For ex- ample, I found in 1955 that it occurred on 30y0 <strong>of</strong> a 2800 tree sample at <strong>the</strong> 1.3 m level on <strong>the</strong> trunk and on 21y0 at base level in mesic deciduous forests. It was most common in closed oak-hickory stands and much rarer in both open, savanna-like oak stands and in heavily shaded maple (Acer saccha- l-urn) climax forests. <strong>Parmelina</strong> aul-ulenta occurs frequently with Parmelia ruderta Acharius and Pseudopal-rnelia capel-ata (L.) <strong>Hale</strong>. In nor<strong>the</strong>rn Wisconsin, an area <strong>of</strong> hardwood- conifer forests more comparable in climate to nor<strong>the</strong>rn Europe, P. aurulenta had an overall fre- quency <strong>of</strong> only Il.670 at <strong>the</strong> 1.3 m level and 1.3% at <strong>the</strong> base. It avoided Pinus spp. and grew almost exclusively on Acel- and Querczis (W. Culberson, 1955). This avoidance <strong>of</strong> Pinus was demonstrated even more graphically by W. Culberson (1958) in an ecological survey exclusively <strong>of</strong> Pinus in North Carolina. Although o<strong>the</strong>rwise common in this state, not a single collection was made on conifers. SPECI~IESS Ex.\xIINED.-United States and Canada: See <strong>Hale</strong> (1958384) for a map <strong>of</strong> distribution based on specimens in various American herbaria. Mexico: Michoacin, Wirth 329 (US). Brazil: Minas Gerais, Eiten 6932 (US). Europe: Russia, Vasiljeva 9 (US). Tanzania: Tanga Province, Santesson 23358, 23359 (Santesson herliarium, US). Union <strong>of</strong> South Africa: Satal, Alniboria 8071, 8079 (LD); Transvaal, Alrnborn 7225 (LD). Madagascar: des .4 bboyes (TUR), Lernaitre (H). Paki-
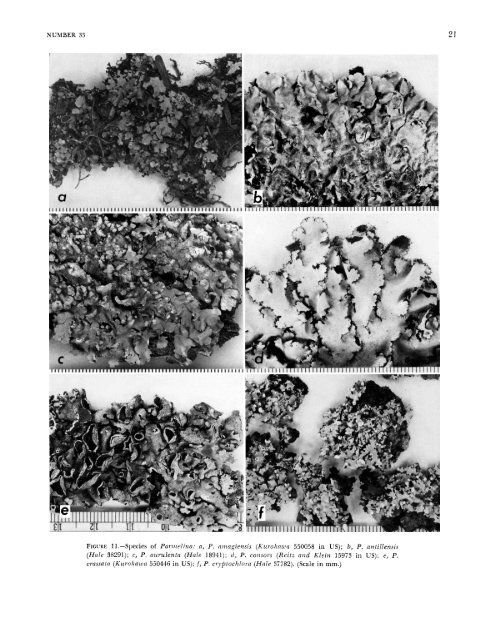
NUMBER 33 FIGURE 11.-Species <strong>of</strong> <strong>Parmelina</strong>: a, P. nmagiensis (Kurokawa 550058 in US); b, P. antillensis (<strong>Hale</strong> 38291); c, P. aurulentn (<strong>Hale</strong> 18941); d, P. consors (Reit- and Klein 15973 in US); e, P. crassutn (Kur<strong>of</strong>mwu 550446 in US); 1, P. cryfitochlora (<strong>Hale</strong> 37782). (Scale in mm.) 21