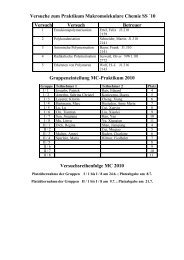
Polymerisation Processes Polymerisation Processes Polyethylene ...
Polymerisation Processes Polymerisation Processes Polyethylene ...
Polymerisation Processes Polymerisation Processes Polyethylene ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
The Nylon revolution 1945<br />
O O<br />
N (CH2 ) N 6 C (CH2 ) C 6 n<br />
H H<br />
Dr. Klaus-Dieter Hungenberg, BASF AG Ludwigshafen Vorlesung <strong>Polymerisation</strong>stechnik, Universität Paderborn, WS 07/08 Polymer processes<br />
Polyamides – base materials<br />
ε-Caprolactam: OH<br />
+6H<br />
(Ni)<br />
OH<br />
H<br />
-2H<br />
O<br />
+H2NOH -H2O N H<br />
Phenol Cyclohexanol Cyclohexanon Cyclohexanon-oxim<br />
Hexamethylene diamine:<br />
Adipic acid:<br />
+6H<br />
(Ni)<br />
Benzene Cyclohexan<br />
H 2C = CH – CH = CH 2<br />
H2C – CH = CH – CH2 CN CN<br />
+CINO<br />
- HCI<br />
(H 2 SO 4 )<br />
H2 C<br />
H2C CO<br />
H2C NH<br />
H2C CH2 ε-Caprolactam:<br />
Dr. Klaus-Dieter Hungenberg, BASF AG Ludwigshafen Vorlesung <strong>Polymerisation</strong>stechnik, Universität Paderborn, WS 07/08 Polymer processes<br />
+Cl 2<br />
+2H<br />
(Ni)<br />
+2NaCN<br />
H2C = CH – CH = CH2 -2NaCl<br />
Cl Cl<br />
NC – (CH2) 4 –CN<br />
+8H<br />
(Ni, NH3 )<br />
H2N – (CH2) 6 –NH2 Adiponitrile Hexamethylendiamin<br />
H2C H2C CH2 C<br />
H2C CH2 H<br />
OH<br />
H2C H2C CH2 C = O<br />
COOH<br />
(CH2 ) 4<br />
H2C CH2 COOH<br />
Cyclohexanol Cyclohexanone Adipic acid<br />
73<br />
75<br />
Polyamides – basic structures<br />
PA 6: Perlon PA6.6: Nylon<br />
O<br />
NH ( CH ) 2 C<br />
NH ( NH<br />
5<br />
CH ) 2 C (<br />
6 CH ) 2 C<br />
4<br />
n<br />
n<br />
PA 6: Caprolactam<br />
PA 11: Undecanlactam<br />
PA 12: Laurinlactam<br />
PA 6.6: Hexamethylendiamine<br />
+ Adipic acid<br />
PA 6.10: Hexamethylendiamien<br />
+ Sebacic acid<br />
PA 4.6: Butandiamine<br />
+ Adipic acid<br />
Dr. Klaus-Dieter Hungenberg, BASF AG Ludwigshafen Vorlesung <strong>Polymerisation</strong>stechnik, Universität Paderborn, WS 07/08 Polymer processes<br />
PA6 – basic reactions<br />
1. Ring opening<br />
NH (CH2 ) CO + H 5 2O H NH (CH2 ) COOH<br />
5<br />
2. Polyaddition<br />
NH (CH2 ) CO + H NH (CH CO OH<br />
5<br />
2 ) 5<br />
3. Polycondensation<br />
n<br />
Dr. Klaus-Dieter Hungenberg, BASF AG Ludwigshafen Vorlesung <strong>Polymerisation</strong>stechnik, Universität Paderborn, WS 07/08 Polymer processes<br />
O<br />
H NH (CH2 ) CO OH<br />
5<br />
H NH (CH2 ) CO OH + H NH (CH CO OH H NH CO OH + H2O 5 2 ) (CH 5 2 ) 5<br />
n<br />
m<br />
n+1<br />
n+m<br />
O<br />
74<br />
76