Toxicity measurements in concentrated water samples - Rivm
Toxicity measurements in concentrated water samples - Rivm
Toxicity measurements in concentrated water samples - Rivm
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
sample with a s<strong>in</strong>gle KD-distillation, the amount of acetone had to be reduced. Therefore, the amount<br />
of XAD should be reduced for about a factor 10 compared to the method which was <strong>in</strong> uses at that<br />
moment, which reduced the costs of the method as well.<br />
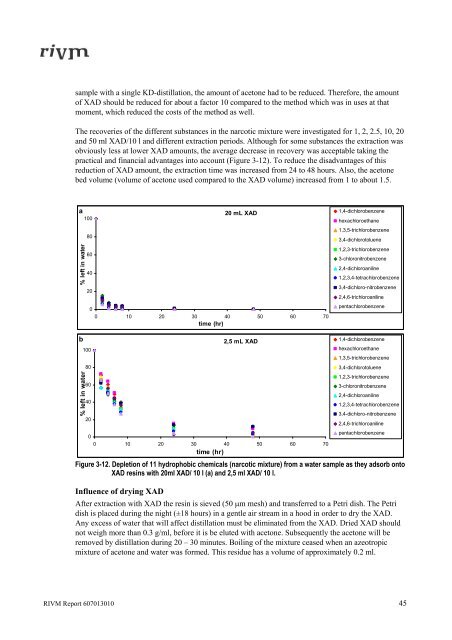
The recoveries of the different substances <strong>in</strong> the narcotic mixture were <strong>in</strong>vestigated for 1, 2, 2.5, 10, 20<br />
and 50 ml XAD/10 l and different extraction periods. Although for some substances the extraction was<br />
obviously less at lower XAD amounts, the average decrease <strong>in</strong> recovery was acceptable tak<strong>in</strong>g the<br />
practical and f<strong>in</strong>ancial advantages <strong>in</strong>to account (Figure 3-12). To reduce the disadvantages of this<br />
reduction of XAD amount, the extraction time was <strong>in</strong>creased from 24 to 48 hours. Also, the acetone<br />
bed volume (volume of acetone used compared to the XAD volume) <strong>in</strong>creased from 1 to about 1.5.<br />
a<br />
% left <strong>in</strong> <strong>water</strong><br />
b<br />
% left <strong>in</strong> <strong>water</strong><br />
100<br />
80<br />
60<br />
40<br />
20<br />
0<br />
100<br />
80<br />
60<br />
40<br />
20<br />
0<br />
20 mL XAD<br />
0 10 20 30 40 50 60 70<br />
time (hr)<br />
2,5 mL XAD<br />
0 10 20 30 40 50 60 70<br />
time (hr)<br />
1,4-dichlorobenzene<br />
hexachloroethane<br />
1,3,5-trichlorobenzene<br />
3,4-dichlorotoluene<br />
1,2,3-trichlorobenzene<br />
3-chloronitrobenzene<br />
2,4-dichloroanil<strong>in</strong>e<br />
1,2,3,4-tetrachlorobenzene<br />
3,4-dichloro-nitrobenzene<br />
2,4,6-trichloroanil<strong>in</strong>e<br />
pentachlorobenzene<br />
1,4-dichlorobenzene<br />
hexachloroethane<br />
1,3,5-trichlorobenzene<br />
3,4-dichlorotoluene<br />
1,2,3-trichlorobenzene<br />
3-chloronitrobenzene<br />
2,4-dichloroanil<strong>in</strong>e<br />
1,2,3,4-tetrachlorobenzene<br />
3,4-dichloro-nitrobenzene<br />
2,4,6-trichloroanil<strong>in</strong>e<br />
pentachlorobenzene<br />
Figure 3-12. Depletion of 11 hydrophobic chemicals (narcotic mixture) from a <strong>water</strong> sample as they adsorb onto<br />
XAD res<strong>in</strong>s with 20ml XAD/ 10 l (a) and 2,5 ml XAD/ 10 l.<br />
Influence of dry<strong>in</strong>g XAD<br />
After extraction with XAD the res<strong>in</strong> is sieved (50 μm mesh) and transferred to a Petri dish. The Petri<br />
dish is placed dur<strong>in</strong>g the night (±18 hours) <strong>in</strong> a gentle air stream <strong>in</strong> a hood <strong>in</strong> order to dry the XAD.<br />
Any excess of <strong>water</strong> that will affect distillation must be elim<strong>in</strong>ated from the XAD. Dried XAD should<br />
not weigh more than 0.3 g/ml, before it is be eluted with acetone. Subsequently the acetone will be<br />
removed by distillation dur<strong>in</strong>g 20 – 30 m<strong>in</strong>utes. Boil<strong>in</strong>g of the mixture ceased when an azeotropic<br />
mixture of acetone and <strong>water</strong> was formed. This residue has a volume of approximately 0.2 ml.<br />
RIVM Report 607013010 45