PHYS01200704032 Debes Ray - Homi Bhabha National Institute
PHYS01200704032 Debes Ray - Homi Bhabha National Institute
PHYS01200704032 Debes Ray - Homi Bhabha National Institute
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Absorbance<br />
Chapter 4: Optimization of the Block Copolymer-mediated Synthesis of Gold Nanoparticles<br />
(b)<br />
2.0<br />
1.6<br />
1.2<br />
2 wt% P85<br />
2 wt% P85 + 0.002 wt% HAuCl4.3H2O<br />
2 wt% P85 + 0.004 wt% HAuCl4.3H2O<br />
2 wt% P85 + 0.006 wt% HAuCl4.3H2O<br />
2 wt% P85 + 0.008 wt% HAuCl4.3H2O<br />
2 wt% P85 + 0.01 wt% HAuCl4.3H2O<br />
2 wt% P85 + 0.012 wt% HAuCl4.3H2O<br />
2 wt% P85 + 0.014 wt% HAuCl4.3H2O<br />
2 wt% P85 + 0.016 wt% HAuCl4.3H2O<br />
2 wt% P85 + 0.018 wt% HAuCl4.3H2O<br />
2 wt% P85 + 0.02 wt% HAuCl4.3H2O<br />
0.8<br />
0.4<br />
0.0<br />
200 400 600 800 1000<br />
Wavelength (nm)<br />
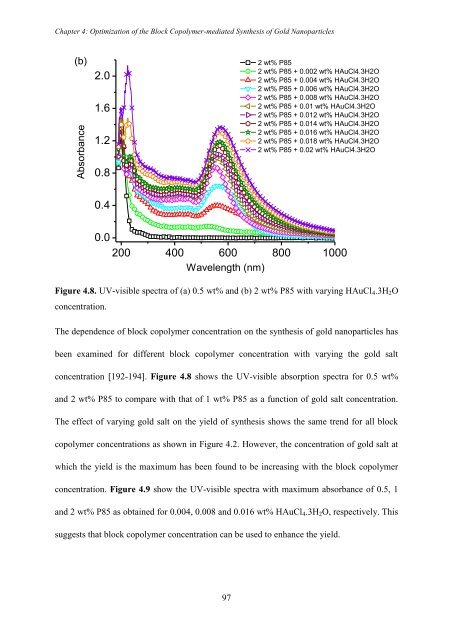
Figure 4.8. UV-visible spectra of (a) 0.5 wt% and (b) 2 wt% P85 with varying HAuCl 4 .3H 2 O<br />
concentration.<br />
The dependence of block copolymer concentration on the synthesis of gold nanoparticles has<br />
been examined for different block copolymer concentration with varying the gold salt<br />
concentration [192-194]. Figure 4.8 shows the UV-visible absorption spectra for 0.5 wt%<br />
and 2 wt% P85 to compare with that of 1 wt% P85 as a function of gold salt concentration.<br />
The effect of varying gold salt on the yield of synthesis shows the same trend for all block<br />
copolymer concentrations as shown in Figure 4.2. However, the concentration of gold salt at<br />
which the yield is the maximum has been found to be increasing with the block copolymer<br />
concentration. Figure 4.9 show the UV-visible spectra with maximum absorbance of 0.5, 1<br />
and 2 wt% P85 as obtained for 0.004, 0.008 and 0.016 wt% HAuCl 4 .3H 2 O, respectively. This<br />
suggests that block copolymer concentration can be used to enhance the yield.<br />
97