Dietary supplements: FDA may have opportunities to expand its use
Dietary supplements: FDA may have opportunities to expand its use
Dietary supplements: FDA may have opportunities to expand its use
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Appendix II: Data on <strong>FDA</strong>’s Consumer<br />
Protection Actions Related <strong>to</strong> <strong>Dietary</strong><br />
Supplements<br />
Data on Warning Letters<br />
Related <strong>to</strong> <strong>Dietary</strong> Supplements<br />
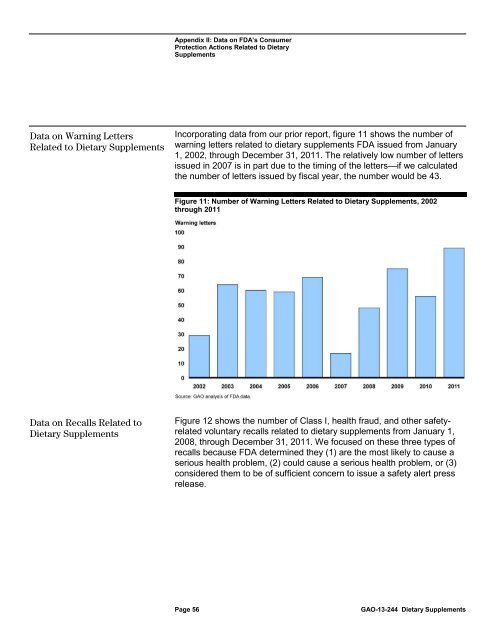
Incorporating data from our prior report, figure 11 shows the number of<br />
warning letters related <strong>to</strong> dietary <strong>supplements</strong> <strong>FDA</strong> issued from January<br />
1, 2002, through December 31, 2011. The relatively low number of letters<br />
issued in 2007 is in part due <strong>to</strong> the timing of the letters—if we calculated<br />
the number of letters issued by fiscal year, the number would be 43.<br />
Figure 11: Number of Warning Letters Related <strong>to</strong> <strong>Dietary</strong> Supplements, 2002<br />
through 2011<br />
Data on Recalls Related <strong>to</strong><br />
<strong>Dietary</strong> Supplements<br />
Figure 12 shows the number of Class I, health fraud, and other safetyrelated<br />
voluntary recalls related <strong>to</strong> dietary <strong>supplements</strong> from January 1,<br />
2008, through December 31, 2011. We foc<strong>use</strong>d on these three types of<br />
recalls beca<strong>use</strong> <strong>FDA</strong> determined they (1) are the most likely <strong>to</strong> ca<strong>use</strong> a<br />
serious health problem, (2) could ca<strong>use</strong> a serious health problem, or (3)<br />
considered them <strong>to</strong> be of sufficient concern <strong>to</strong> issue a safety alert press<br />
release.<br />
Page 56<br />
GAO-13-244 <strong>Dietary</strong> Supplements