Combinatorial and High-Throughput Screening of Materials ...
Combinatorial and High-Throughput Screening of Materials ...
Combinatorial and High-Throughput Screening of Materials ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
ACS <strong>Combinatorial</strong> Science<br />
REVIEW<br />
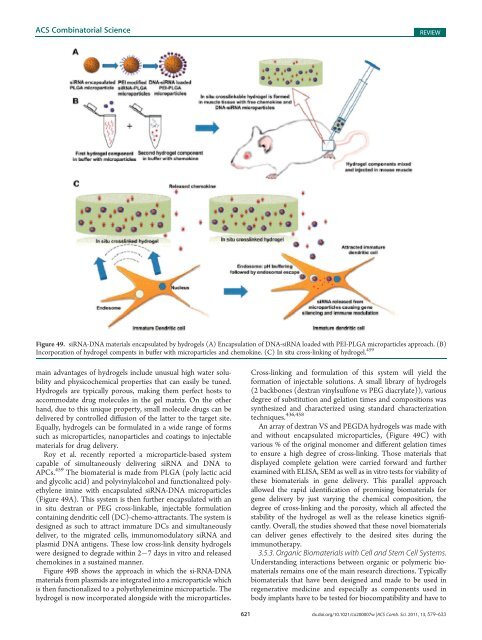
Figure 49. siRNA-DNA materials encapsulated by hydrogels (A) Encapsulation <strong>of</strong> DNA-siRNA loaded with PEI-PLGA microparticles approach. (B)<br />
Incorporation <strong>of</strong> hydrogel compents in buffer with microparticles <strong>and</strong> chemokine. (C) In situ cross-linking <strong>of</strong> hydrogel. 459<br />
main advantages <strong>of</strong> hydrogels include unusual high water solubility<br />
<strong>and</strong> physicochemical properties that can easily be tuned.<br />
Hydrogels are typically porous, making them perfect hosts to<br />
accommodate drug molecules in the gel matrix. On the other<br />
h<strong>and</strong>, due to this unique property, small molecule drugs can be<br />
delivered by controlled diffusion <strong>of</strong> the latter to the target site.<br />
Equally, hydrogels can be formulated in a wide range <strong>of</strong> forms<br />
such as microparticles, nanoparticles <strong>and</strong> coatings to injectable<br />
materials for drug delivery.<br />
Roy et al. recently reported a microparticle-based system<br />
capable <strong>of</strong> simultaneously delivering siRNA <strong>and</strong> DNA to<br />
APCs. 459 The biomaterial is made from PLGA (poly lactic acid<br />
<strong>and</strong> glycolic acid) <strong>and</strong> polyvinylalcohol <strong>and</strong> functionalized polyethylene<br />
imine with encapsulated siRNA-DNA microparticles<br />
(Figure 49A). This system is then further encapsulated with an<br />
in situ dextran or PEG cross-linkable, injectable formulation<br />
containing dendritic cell (DC)-chemo-attractants. The system is<br />
designed as such to attract immature DCs <strong>and</strong> simultaneously<br />
deliver, to the migrated cells, immunomodulatory siRNA <strong>and</strong><br />
plasmid DNA antigens. These low cross-link density hydrogels<br />
were designed to degrade within 2 7 days in vitro <strong>and</strong> released<br />
chemokines in a sustained manner.<br />
Figure 49B shows the approach in which the si-RNA-DNA<br />
materials from plasmids are integrated into a microparticle which<br />
is then functionalized to a polyethyleneimine microparticle. The<br />
hydrogel is now incorporated alongside with the microparticles.<br />
Cross-linking <strong>and</strong> formulation <strong>of</strong> this system will yield the<br />
formation <strong>of</strong> injectable solutions. A small library <strong>of</strong> hydrogels<br />
(2 backbones (dextran vinylsulfone vs PEG diacrylate)), various<br />
degree <strong>of</strong> substitution <strong>and</strong> gelation times <strong>and</strong> compositions was<br />
synthesized <strong>and</strong> characterized using st<strong>and</strong>ard characterization<br />
techniques. 436,458<br />
An array <strong>of</strong> dextran VS <strong>and</strong> PEGDA hydrogels was made with<br />
<strong>and</strong> without encapsulated microparticles, (Figure 49C) with<br />
various % <strong>of</strong> the original monomer <strong>and</strong> different gelation times<br />
to ensure a high degree <strong>of</strong> cross-linking. Those materials that<br />
displayed complete gelation were carried forward <strong>and</strong> further<br />
examined with ELISA, SEM as well as in vitro tests for viability <strong>of</strong><br />
these biomaterials in gene delivery. This parallel approach<br />
allowed the rapid identification <strong>of</strong> promising biomaterials for<br />
gene delivery by just varying the chemical composition, the<br />
degree <strong>of</strong> cross-linking <strong>and</strong> the porosity, which all affected the<br />
stability <strong>of</strong> the hydrogel as well as the release kinetics significantly.<br />
Overall, the studies showed that these novel biomaterials<br />
can deliver genes effectively to the desired sites during the<br />
immunotherapy.<br />
3.5.3. Organic Biomaterials with Cell <strong>and</strong> Stem Cell Systems.<br />
Underst<strong>and</strong>ing interactions between organic or polymeric biomaterials<br />
remains one <strong>of</strong> the main research directions. Typically<br />
biomaterials that have been designed <strong>and</strong> made to be used in<br />
regenerative medicine <strong>and</strong> especially as components used in<br />
body implants have to be tested for biocompatibility <strong>and</strong> have to<br />
621 dx.doi.org/10.1021/co200007w |ACS Comb. Sci. 2011, 13, 579–633