Pharmacology
Pharmacology
Pharmacology
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
<strong>Pharmacology</strong><br />
from the ancient Greek word for drug pharmakon<br />
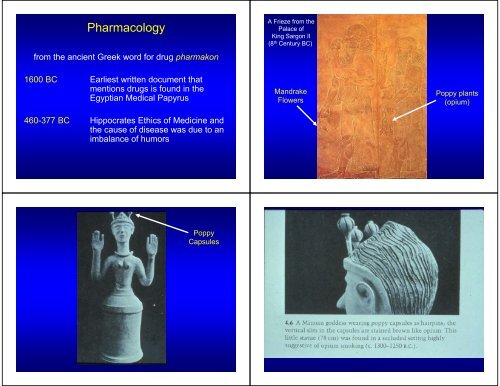
A Frieze from the<br />
Palace of<br />
King Sargon II<br />
(8 th Century BC)<br />
1600 BC Earliest written document that<br />
mentions drugs is found in the<br />
Egyptian Medical Papyrus<br />
460-377 BC Hippocrates Ethics of Medicine and<br />
the cause of disease was due to an<br />
imbalance of humors<br />
Mandrake<br />
Flowers<br />
Poppy plants<br />
(opium)<br />
Poppy<br />
Capsules
57 AD Dioscorides - Materia Medica over 500 plants and<br />
remedies<br />
130-201 Galen - Theory of disease<br />
1493-1541 Paracelsus – Grandfather of <strong>Pharmacology</strong>, “All<br />
things are poisons…..depends upon the dose”<br />
1805 Serturner – Isolated Morphine<br />
1909 Ehrlich - Chemotherapy<br />
1935 Domagk – discover first sulfonamide<br />
1950’s Beyer – development of thiazides & others<br />
<strong>Pharmacology</strong><br />
The study of substances (drugs) on living systems<br />
Pharmacokinetics deals with the absorption, distribution, biotransformation and<br />
excretion of drugs<br />
Pharmacodynamics deals with the study of the biochemical and physiological<br />
effects of drugs and their mechanism of action<br />
Toxicology deals with adverse effects of drugs and chemicals<br />
Pharmacotherapeutics the use of drugs in the prevention and treatment of<br />
disease<br />
Virtually all drugs produce more than one effect yet one effect that<br />
predominates over a particular dose range is referred to the therapeutic<br />
window.<br />
1960/70’s<br />
development of Propanolol and Cimetidine<br />
Drugs are used to prevent, diagnose and/or treat disease. Drugs modify<br />
physiological processes-they DO NOT create new processes or effects<br />
General Drug Mechanism<br />
Drugs are used to correct defects in physiology<br />
Deficiency of some essential component – replacement therapy<br />
Most Drugs act at Receptors to produce and effect<br />
In order for drugs to interact “bind” with receptors they should have:<br />
-Shape<br />
-Size<br />
-Charge<br />
-Atomic Composition<br />
Affinity<br />
Excess Action of some normal or even essential ingredient –<br />
chemical antagonist or exogenous substances<br />
The Physiochemical Environment of the specific part of the body<br />
may be altered by a drug – “nonspecific”
Dose Response<br />
Lock & Key Theory of<br />
Drug Receptor Interaction<br />
Drug-Receptor<br />
Complex<br />
+ -<br />
Drug<br />
-<br />
+<br />
Receptor<br />
Lock & Key Theory of<br />
Drug Receptor Interaction<br />
Drug Receptor - Affinity<br />
How well a drug binds to a receptor is dictated<br />
by the affinity of the drug for the receptor<br />
Drug-Receptor<br />
Complex<br />
Dynamic<br />
Drug<br />
D + R<br />
k +1<br />
k -1<br />
DR<br />
+ -<br />
-<br />
+<br />
Receptor<br />
(Dissociation Constant)<br />
K A = k-1<br />
k +1<br />
Low K A =<br />
High Affinity
Receptors<br />
The majority of all Receptors are proteins<br />
Receptors bind with drugs & bring about<br />
change in cell function<br />
Evidence for Receptors<br />
1. Extreme Potency/Efficacy of Drugs<br />
2. Chemical Selectivity (i.e., similar molecules<br />
produce similar effects)<br />
Evidence for Receptors<br />
1. Extreme Potency/Efficacy of Some Drugs<br />
2. Chemical Selectivity (i.e., similar molecules<br />
produce similar effects)<br />
3. Stereoselectivity
L-Epinephrine<br />
D-Epinephrine<br />
L-Epinephrine<br />
D-Epinephrine<br />
OH<br />
H<br />
H<br />
CH 3<br />
OH<br />
OH<br />
H<br />
CH 3<br />
OH C C N<br />
OH<br />
H<br />
H<br />
OH C C N<br />
H<br />
H<br />
H<br />
Stereo-Selectivity<br />
OH<br />
H<br />
H<br />
CH 3<br />
OH<br />
OH<br />
H<br />
CH 3<br />
OH C C N<br />
OH C C N<br />
OH<br />
H<br />
H<br />
H<br />
H<br />
H<br />
Intracellular Effects<br />
Evidence for Receptors<br />
Targets for Drug Action<br />
1. Extreme Potency/Efficacy of Some Drugs<br />
2. Chemical Selectivity (i.e., similar molecules<br />
produce similar effects)<br />
3. Stereoselectivity<br />
4. Competitive Antagonists<br />
5. Cloning and Expression of Receptors
Drug<br />
G-protein Coupled Receptors<br />
Drug<br />
NT<br />
NT<br />
AC<br />
GDP<br />
G-protein Coupled Receptors<br />
G-protein Coupled Receptors<br />
NT<br />
GIRK<br />
NT<br />
K +<br />
GIRK<br />
AC<br />
AC<br />
NT NT<br />
NT NT<br />
βγ<br />
α βγ<br />
GDP<br />
α<br />
GTP<br />
K +<br />
GTP<br />
GDP
G-protein Coupled Receptors<br />
G-protein Coupled Receptors<br />
NT<br />
K +<br />
NT<br />
K +<br />
AC<br />
AC<br />
NT NT<br />
K +<br />
NT NT<br />
α<br />
GTP<br />
cAMP<br />
K +<br />
α<br />
GTP<br />
ATP<br />
GDP<br />
GDP<br />
amplification<br />
PKA<br />
cREB<br />
G-protein Coupled Receptors<br />
NT<br />
AC<br />
NT NT<br />
α<br />
GDP<br />
GTP<br />
P<br />
βγ<br />
PDE<br />
cAMP<br />
5’AMP<br />
amplification<br />
PKA<br />
cREB
G q -protein Coupled Receptors<br />
Cont. G q -protein Coupled Receptors<br />
G q Coupling<br />
(G q -protein)<br />
CAM<br />
kinase<br />
Ca 2+<br />
Ca 2+<br />
PKC<br />
PIP 2<br />
PLC<br />
IP3<br />
DAG
Tyrosine Kinase Receptors<br />
Kinase<br />
cascade<br />
2 nd Messenger linked Receptors (metabotropic)
Dose Response Curves<br />
Dose Response Curves<br />
Propionylcholine<br />
100 Acetylcholine<br />
Acetylcholine<br />
100<br />
% Response<br />
75<br />
50<br />
25<br />
Propionylcholine<br />
75<br />
50<br />
25<br />
0<br />
0.0 0.2 0.4 0.6 0.8 1.0<br />
Dose ( μg/ml)<br />
arithmetic scale<br />
0<br />
0.001 0.01 1.0 10.0<br />
Dose ( μg/ml)<br />
logarithmic scale<br />
% Response<br />
100<br />
75<br />
50<br />
25<br />
Relevant Data on a Dose<br />
Response Curve<br />
80%<br />
20%<br />
Three Different Types of Dose<br />
Response Curves<br />
0<br />
0.001 0.01 1.0 10.0<br />
Log Dose (ug/ml)<br />
ED 50 = 0.05ug (x- intercept)
Quantal Dose Response Curve<br />
Graded Dose Response Curve<br />
100<br />
100<br />
# Responding<br />
75<br />
50<br />
25<br />
Variable:<br />
Dose<br />
Constant:<br />
Time<br />
Response<br />
% Response<br />
75<br />
50<br />
25<br />
Variable:<br />
Dose<br />
Response<br />
Constant:<br />
Time<br />
0<br />
0.001 0.01 1.0 10.0<br />
0<br />
0.001 0.01 1.0 10.0<br />
Log Dose ( μg/ml)<br />
Log Dose ( μg/ml)<br />
Time Action Curve<br />
Potency vs. Efficacy<br />
% Response<br />
100<br />
80<br />
60<br />
40<br />
20<br />
1 mg/kg<br />
3 mg/kg<br />
10 mg/kg<br />
Variable:<br />
Dose<br />
Response<br />
Time<br />
% Response<br />
100<br />
75<br />
50<br />
25<br />
0<br />
Efficacy A<br />
B<br />
Potency<br />
0.001 0.01 1.0 10.0<br />
C<br />
0<br />
0 15 30 45 60 75 90 105 120<br />
Log Dose (ug/ml)<br />
Time (min)
Dose Response Curves<br />
Dose Response Curves<br />
% Response<br />
100<br />
75<br />
50<br />
25<br />
0<br />
Partial Agonist<br />
B<br />
C<br />
A<br />
Full Agonist<br />
Antagonist<br />
Potency: Left/Right shift of the dose response curve,<br />
Amount of drug required to produce a response<br />
Efficacy: The height of the dose response curve,<br />
-25<br />
D<br />
Inverse Agonist<br />
Ability of a drug to induce a maximal response<br />
0.001 0.01 1.0 10.0<br />
Log Dose (ug/ml)<br />
Dose Response Curves<br />
Does and Antagonist have Affinity<br />
Does and Antagonist have Efficacy<br />
Does and Antagonist have Potency<br />
YES<br />
NO<br />
YES<br />
Which has higher Efficacy<br />
Partial agonist<br />
Full agonist<br />
Partial agonist<br />
Which has higher Potency<br />
Full agonist<br />
Can’t tell without looking at a DRC
Shift in Potency<br />
No loss of Efficacy<br />
Competitive Antagonist<br />
Shift in Potency<br />
loss of Efficacy<br />
Drugs Often Result in<br />
Several Biological<br />
Responses
Drugs Produce More Than One Effect<br />
Drugs Produce More Than One Effect<br />
% Response<br />
100<br />
75<br />
50<br />
25<br />
constipation<br />
antitusive<br />
analgesia<br />
resp. depression<br />
death<br />
% Response<br />
100<br />
75<br />
50<br />
25<br />
Therapeutic Index “window”<br />
LD 50<br />
ED 50<br />
=<br />
50<br />
2<br />
= 25<br />
analgesia<br />
death<br />
0<br />
0.001 0.01 1.0 10.0<br />
100.0 1000.0<br />
0<br />
0.001 0.01 1.0 10.0<br />
100.0 1000.0<br />
Log Morphine (mg/kg)<br />
ED 50 (2mg/kg)<br />
LD 50 (50mg/kg)<br />
Log Morphine (mg/kg)<br />
Response According to Age Sex Health<br />
% Response<br />
100<br />
80<br />
60<br />
40<br />
20<br />
Teenager Female Lance Armstrong<br />
Middle Male Todd Vanderah Age<br />
Elderly Dick Cheney<br />
0<br />
1 3 10 30 100 300<br />
Log [Drug]
Review<br />
Agonists: Affinity, Selectivity, Potency and Efficacy<br />
Antagonists: Affinity, Selectivity & Potency<br />
Competitive Antagonists: Decrease Potency<br />
Non-Competitive Antagonists: Decrease Potency<br />
Decrease Efficacy<br />
Drugs often produce more than one effect<br />
Drug Response depends on the tissue it acts on<br />
Review<br />
Categories of Receptors<br />
- Ion Channels (ionotropic)<br />
-Enzymes<br />
- Transporters<br />
-2 nd Messenger Receptors<br />
- Nuclear Receptors<br />
- Kinase receptors<br />
-G-proteins<br />
Activation of<br />
G-proteins<br />
Leads to<br />
G αs<br />
G αi<br />
G αq<br />
Decrease cAMP and PKA activity<br />
Activate the Kinase Cascade<br />
Increase cAMP and PKA activity<br />
PLA 2<br />
Tyrosine Kinase Receptors<br />
Adenylate Cyclase<br />
Increase Arachidonic Acid Release<br />
Increase IP3 and DAG<br />
Increase PKC and CAM kinase activity<br />
Decrease IP3 and DAG
Pain and Opioid Mechanisms<br />
Pain<br />
• Most common reason for seeking medical care<br />
• Everyone experiences pain…30% of Americans will<br />
suffer from chronic pain<br />
• Treatment of pain improves outcome;<br />
• Chronic pain is itself a disease which can and should<br />
be treated.<br />
Congenital Insensitivity to Pain
C & Aδ Fibers<br />
Pain Pathways<br />
Somatosensory<br />
Cortex<br />
Thalamus<br />
From<br />
Brain<br />
To<br />
Brain<br />
Spinal<br />
Cord<br />
Neural Steps in the Processing of Pain Signals<br />
1. Transduction - Noxious stimuli → electrical signals<br />
2. Transmission - Neural events from the periphery to the<br />
cortex<br />
3. Modulation - Nervous system can selectively inhibit or<br />
enhance pain signals<br />
4. Perception - subjective interpretation by the cortex<br />
Sensory component<br />
Affective component<br />
Nociceptive Transmission Pathway<br />
Sensory<br />
Receptor Periphery CNS<br />
Nociceptors:<br />
• C-polymodal<br />
small, non-myelinated<br />
• Aδ-mechanical<br />
thermal<br />
Medium, thinly-myelinated<br />
Spinal<br />
cord<br />
DRG<br />
Thalamus
C-Polymodal Nociceptors<br />
Pressure<br />
Nociceptors (C & Aδ fibers)<br />
Free nerve endings that do NOT adapt<br />
Heat<br />
Respond to many modalities<br />
Action Physical: heat, cold, force<br />
Potentials Chemical: capsaicin, protons, bradykinin, ATP, PGE 2<br />
Found Heat throughout body:<br />
Stimuli<br />
skin, muscle, joint, viscera, bone<br />
48 ºC<br />
Chemical<br />
Some are unresponsive “silent”<br />
only respond after injury or inflammation<br />
TRPV1 receptor<br />
ion channel<br />
Na +<br />
Ca +2<br />
outer<br />
Inner<br />
Heat<br />
Activation<br />
Peripheral Nociceptor Activation<br />
↓pH<br />
Receptor Potential<br />
Prostaglandins (PG)<br />
K +<br />
Bradykinin (BK)<br />
Capsaicin<br />
Used to identify nociceptors<br />
Trigger Zone
Peripheral Nociceptor Activation<br />
“Axon Reflex”<br />
Peripheral Nociceptor Activation<br />
“Recruitment of Surrounding Nociceptors”<br />
H<br />
mast cell<br />
PG SP<br />
H<br />
PG<br />
SP<br />
PG<br />
SP<br />
PG<br />
platelet<br />
BK<br />
Neuronal recruitment<br />
Trigger Zone<br />
Peripheral Nociceptor Activation<br />
“Actions of Pain Relievers”<br />
Sensitized Nociceptors<br />
Allodynia<br />
Hyperalgesia<br />
Opioids<br />
Neuronal recruitment<br />
H<br />
PG<br />
SP<br />
PG<br />
Opioids<br />
1) Opioids act to hyperpolarize the nociceptor<br />
2) Opioids act to reduce inflammatory mediators<br />
From immune cells (also the site of NSAIDs)<br />
Subjective Pain Intensity<br />
6<br />
5<br />
4<br />
3<br />
2<br />
1<br />
41 43 45 47 49<br />
Stimulus Temperature ( o C)<br />
After<br />
injury<br />
Naive
C and Aδ Nociceptor Mediated Pain<br />
C-fiber<br />
Touch fibers<br />
DRG<br />
Dorsal<br />
Columns<br />
Spinothalamic<br />
Tract<br />
Periphery<br />
Spinal Cord<br />
Pain Fibers<br />
Aδ fiber<br />
Lisauer’s tract<br />
I<br />
Pain<br />
intensity<br />
First<br />
pain<br />
Second<br />
pain<br />
laminae<br />
II<br />
IV<br />
V<br />
Time<br />
Ventral<br />
Roots<br />
Burn<br />
Spinothalamic<br />
Projection to<br />
Thalamus<br />
Opioids Inhibit Noxious Input<br />
Nociceptive<br />
Input<br />
Flare<br />
ENK<br />
Ca ++<br />
Substantia<br />
gelatinosa<br />
Spinal<br />
Cord<br />
μ Opioid Receptors<br />
Pain Transmitters:<br />
Glutamate<br />
Substance P<br />
Dorsal Horn,<br />
Spinal Cord<br />
EPSP
Spinothalamic<br />
Projection to<br />
Thalamus<br />
Opioids Inhibit Noxious Input<br />
ENK<br />
Morphine / Fentanyl<br />
Nociceptive<br />
Input<br />
Ca ++ Conductance<br />
Substantia<br />
gelatinosa<br />
K + efflux (hyperpolarize)<br />
Afferent Pathways<br />
Underlying The Sensation<br />
of Pain<br />
MIDBRAIN<br />
MEDULLA<br />
THALAMUS<br />
STT<br />
Activation of μ<br />
Opioid Receptors<br />
Morphine / Fentanyl<br />
Pain Transmitters:<br />
Substance P<br />
Glutamate<br />
CGRP<br />
K + efflux (hyperpolarize)<br />
DRG<br />
Glutamate<br />
Substance P<br />
2º<br />
SPINAL CORD<br />
Descending<br />
Pain<br />
Pathways<br />
To thalamus<br />
Rostral<br />
midbrain<br />
From hypothalamus,<br />
amygdala, cortex<br />
Periaqueductal<br />
gray<br />
Descending Modulation of Spinal Nociceptive Input<br />
Opioid Disinhibition<br />
PAG<br />
CP/RM<br />
GABA A<br />
CL -<br />
GABA<br />
Caudal Pons<br />
Rostral Medulla<br />
Nucleus<br />
Raphe magnus<br />
From pain<br />
nociceptors<br />
Dorsolateralfuniculus<br />
(DLF)<br />
(Hyperpolarize)
Descending Modulation of Spinal Nociceptive Input<br />
PAG<br />
Opioid Disinhibition<br />
Morphine<br />
(Hyperpolarize)<br />
CP/RM<br />
Release<br />
5HT & NE<br />
Enk/End<br />
GABA A<br />
CL -<br />
(Hyperpolarize)<br />
X<br />
GABA<br />
release<br />
GABA<br />
K +<br />
DLF<br />
PAG<br />
CP/RM<br />
Release<br />
Enkephalins / Endorphins<br />
Serotonin / Norepinephrine<br />
Opioid Receptors<br />
• Three cloned opioid receptors<br />
– mu, delta and kappa<br />
• Opioid receptors are coupled to the G i /G o class of<br />
G-protein coupled receptors<br />
• Opioid agonist cause inhibition of adenylyl cyclase<br />
and decrease cAMP formation<br />
• Selective agonists:<br />
– Fentanyl<br />
– Morphine<br />
– Meperidine<br />
• Selective antagonists:<br />
– β-FNA<br />
N<br />
OH<br />
μ Opioid Receptor<br />
Mu<br />
• Most of the currently available opioids work<br />
through the mu opioid receptor<br />
HO<br />
O<br />
N<br />
H<br />
O<br />
O<br />
O
δ Opioid Receptor<br />
κ Opioid Receptor<br />
• Selective agonists:<br />
– DPDPE<br />
– SNC80<br />
– Deltorphin II<br />
• Selective antagonist:<br />
– Naltrindole<br />
• Selective agonists:<br />
– Butorphanol<br />
– Ketocyclazocine<br />
• Selective antagonist:<br />
– nor-BNI<br />
N<br />
OH<br />
N<br />
OH<br />
N<br />
HO<br />
HO<br />
O<br />
N<br />
H<br />
HO<br />
O<br />
N<br />
H<br />
O<br />
OH<br />
Mammalian Endogenous Opioid Peptides<br />
Opioid Receptor <strong>Pharmacology</strong><br />
Precursor Endogenous peptide Amino acid sequence<br />
Pro-opiomelanocortin β-Endorphin YGGFMTSEKSQTPLVTLFKNAIIKNAYKKGE<br />
Pro-enkephalin [Met]enkephalin YGGFM<br />
[Leu]enkephalin<br />
YGGFL<br />
YGGFMRF<br />
YGGFMRGL<br />
Metorphamide<br />
YGGFMRRV-NH2<br />
Pro-dynorphin Dynorphin A YGGFLRRIRPKLKWDNQ<br />
Dynorphin A(1-8)<br />
YGGFLRRI<br />
Dynorphin B<br />
YGGFLRRQFKVVT<br />
α-neoendorphin<br />
YGGFLRKYPK<br />
β-neoendorphin<br />
YGGFLRKYP<br />
Pro-nociceptin / OFQ Nociceptin FGGFTGARKSARKLANQ<br />
Pro-endomorphin* Endomorphin-1 YPWF-NH2<br />
Endomorphin-2<br />
YPFF-NH2<br />
•Different pharmacological effects are mediated by<br />
the different receptors<br />
CNS<br />
CNS<br />
Gut<br />
CNS<br />
CNS<br />
CNS<br />
CNS<br />
Pharmacological Effects of Opioid Receptor<br />
Activation<br />
Effect Mu (μ) Delta (δ) Kappa (κ)<br />
Analgesia +++ +++ +<br />
Respiratory Depression +++ 0 +<br />
Constipation +++ + 0<br />
Prolactin Release +++ 0 +<br />
Euphoria +++ 0<br />
Dependence Potential +++ + 0<br />
Dysphoria + +++<br />
*Presumed to exist, awaiting discovery
Main Effects of Opioids at Therapeutic Doses<br />
Physical Dependence<br />
Desirable Undesirable Mixed Desirability<br />
Analgesia Nausea, vomiting Sedation<br />
Relief of anxiety Urinary retention Euphoria<br />
Dysphoria<br />
Cough suppression<br />
Mental clouding ↓ Bowel motility<br />
Tolerance<br />
Drug dependence syndrome<br />
Respiratory depression<br />
Postoperative ileus<br />
Biliary spasm<br />
• Prolonged exposure to an opioid agonist can<br />
produce adaptations at the cellular and systems<br />
levels which oppose the drug effect<br />
– the patient has become physically dependent on the<br />
drug<br />
• physical dependence does not equal addiction!!!<br />
• The discontinuation or sudden reduction in drug<br />
levels, or the administration of an antagonist, can<br />
lead to a withdrawal syndrome<br />
Symptoms and Signs of Opioid Withdrawal<br />
Addiction Liability<br />
Symptoms<br />
restlessness<br />
irritability<br />
increased sensitivity to pain<br />
nausea<br />
abdominal cramps<br />
myalgia<br />
dysphoria<br />
insominia<br />
anxiety<br />
drug craving<br />
Signs<br />
pupillary dilation<br />
sweating<br />
tachycardia<br />
vomiting<br />
diarrhea<br />
hypertension<br />
yawning<br />
fever<br />
rhinorrhea<br />
piloerection<br />
• Studies in the 1920s and 1950s were influential in<br />
setting opioid analgesia policy in the later part of<br />
the 20 th century<br />
– chronic opioid therapy leads to addiction<br />
– Kathleen Foley quote<br />
• These studies had methodological flaws and more<br />
recent reports have suggested that the risk of<br />
iatrogenic opioid addiction is very low<br />
– the incidence of addiction in chronic pain patients<br />
parallels the lifetime prevalence rates in the general<br />
population (Fishbain et al., 1992)
Addiction Liability<br />
Dopaminergic Disinhibition<br />
VTA<br />
Mesolimbic & Mesocortical<br />
Dopaminergic Pathway<br />
Mesolimbic & Mesocortical<br />
Dopaminergic Pathway<br />
Dopamine<br />
Release<br />
GABA A<br />
CL -<br />
(Hyperpolarize)<br />
GABA A<br />
CL -<br />
(Hyperpolarize)<br />
(Hyperpolarize)<br />
Morphine<br />
X<br />
GABA<br />
release<br />
GABA<br />
GABA<br />
K +<br />
Respiratory Depression<br />
• Respiratory depression is the most lifethreatening<br />
of the side-effects associated with<br />
opioid analgesics<br />
– major cause of fatal opioid overdoses<br />
• Opioid receptors are located on chemoreceptors<br />
and ventral respiratory group neurons in the<br />
medulla<br />
– diminished sensitivity to changes in O 2 and CO 2 levels<br />
– changes in tidal volume and frequency of respiration<br />
Constipation<br />
• One of the biggest concerns with chronic opioid<br />
administration is severe constipation<br />
– occurs in almost all patients receiving chronic opioids<br />
• In many cases, constipation is the primary reason<br />
for the patient discontinuing opioid therapy<br />
– development of opioid antagonists that do not cross<br />
the blood-brain barrier<br />
Oral<br />
Peristalsis Produced by Coordinated<br />
Contraction and Relaxation of Muscle Coats<br />
Longitudinal muscle<br />
*****<br />
*****<br />
Bolus<br />
*****<br />
*****<br />
Myenteric plexus<br />
(Auerbach’s)<br />
***** *****<br />
***** *****<br />
Anal<br />
Contracted<br />
Relaxed<br />
Circular muscle<br />
Submucous plexus<br />
(Meissner’s)
Opioids and Intestinal Motility<br />
Segmenting Contractions<br />
Normal<br />
Reduced Contractions<br />
{<br />
Diarrhea<br />
Propulsive Contractions<br />
Segmenting Contractions<br />
Opioids<br />
Loperamide (Imodium)<br />
Normal<br />
Flow<br />
Increased<br />
Flow<br />
Increased<br />
Flow<br />
Decreased<br />
Flow<br />
Drug Interactions<br />
• Many other CNS depressants can act in an additive or<br />
synergistic manner with opioid analgesics<br />
– benzodiazepines<br />
–barbiturates<br />
–alcohol<br />
– antipsychotics<br />
• MAO inhibitors with opioids may produce hyperpyrexic<br />
coma and hypertension<br />
– especially with meperidine<br />
• Stimulants have sometimes been used to counteract the<br />
CNS depressant effects of opioids<br />
Morphine<br />
Methadone<br />
• Prototypical mu opioid agonist<br />
– still isolated from raw opium<br />
• Multiple preparations available<br />
–parenteral<br />
– oral and sustained release (MS Contin)<br />
–rectal<br />
• Indicated for moderate to severe pain<br />
• Full opioid agonist<br />
– efficacy similar to morphine<br />
– good oral bioavailability<br />
– long half-life and duration of action<br />
• The compound has antagonist actions at the<br />
NMDA receptor complex<br />
– decreased development of tolerance and physical<br />
dependence<br />
– role in treatment of neuropathic pain<br />
• Used in the treatment of heroin addiction
Fentanyl and Analogues<br />
Meperidine<br />
• Relatively selective mu opioid agonists<br />
– typically more potent than morphine<br />
– shorter duration of action (e.g., remifentanil)<br />
– high efficacy compounds<br />
• Fentanyl can be given by various routes<br />
– transdermal patch<br />
– fentanyl lozenge (lollipop)<br />
• Moderate efficacy opioid agonist<br />
– used for moderate pain<br />
• The compound has antimuscarinic effects<br />
– cardiovascular effects possible<br />
– limited constriction of the pupil<br />
• Accumulation of metabolite (normeperidine) can<br />
produce seizures<br />
– caution with high doses and in patients with renal<br />
insufficiency<br />
Codeine<br />
Oxycodone<br />
• Codeine is a weak opioid analgesic<br />
– has limited affinity for opioid receptors<br />
– considered a prodrug<br />
• Needs to be demethylated to morphine<br />
– approximately 10% of Caucasians lack the cytochrome<br />
P450 isozyme (CYP 2D6)<br />
– these patients will get no relief from even high doses<br />
of codeine<br />
• Opioid agonist with moderate efficacy<br />
• Recent controversy with this drug<br />
– sustained release preparation-OxyContin<br />
– pellets can be crushed and a solution injected<br />
• Purdue Pharma has tried to reformulate the<br />
compound with naloxone<br />
– oral administration would still be effective<br />
– parenteral administration would result in blockade of<br />
opioid actions
Propoxyphene<br />
Diphenoxylate/Loperamide<br />
• Very weak opioid agonist<br />
– used in combination with aspirin and<br />
acetaminophen<br />
• Limited clinical utility<br />
• Diphenoxylate is a weak opioid agonist with low<br />
abuse liability<br />
– atropine added as a combination product for<br />
treatment of diarrhea and to discourage abuse<br />
• Loperamide is a weak opioid agonist that does not<br />
readily cross the blood-brain barrier<br />
– low abuse liability<br />
– common OTC antidiarrheal medication<br />
Mixed Opioid Agonists/Antagonists<br />
Tramadol<br />
• These compounds are characterized by different<br />
actions on each of the opioid receptors<br />
–nalbuphine-kappa agonist/mu antagonist<br />
–buprenorphine-partial mu agonist<br />
– pentazocine-kappa agonist, weak mu agonist<br />
• These agents are contraindicated in opioid<br />
dependent patients<br />
– severe withdrawal syndrome may develop<br />
• Novel analgesic action<br />
– weak mu opioid agonist<br />
– blocks reuptake of serotonin<br />
– blocks reuptake of norepinephrine<br />
• Marketed as a compound with efficacy similar to<br />
codeine with less side-effects<br />
– no addiction liability<br />
– less GI effects<br />
– efficacy in neuropathic pain states<br />
–expensive
Naloxone<br />
Naltrexone<br />
• Prototypical opioid antagonist<br />
• Used to treat opioid overdose<br />
– low oral bioavailability<br />
– short duration of action<br />
• Will precipitate a severe withdrawal syndrome in<br />
opioid dependent patients<br />
• New evidence suggests that this compound has<br />
inverse agonist actions in opioid dependent states<br />
– increase in withdrawal symptoms<br />
• Prototypical opioid antagonist<br />
• Used to treat heroin addicts<br />
– good oral bioavailability<br />
– long duration of action<br />
• New evidence suggests that this compound has<br />
inverse agonist actions in opioid dependent states<br />
– role in patient compliance<br />
• Approved for the treatment of alcoholism<br />
– decreases binge type drinking<br />
Conclusions<br />
Tucson, Arizona Pain Group<br />
• Pain is mediated by nociceptors and perceived by the<br />
brain.<br />
• Multiple sites where opioids work to relieve pain.<br />
• Opioids produce multiple effects.<br />
• Opioids are abused for their euphoric effects by<br />
indirectly causing an increase in dopamine release.