A Self-Assessment Guide for Health Care Organizations - IFC
A Self-Assessment Guide for Health Care Organizations - IFC
A Self-Assessment Guide for Health Care Organizations - IFC
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
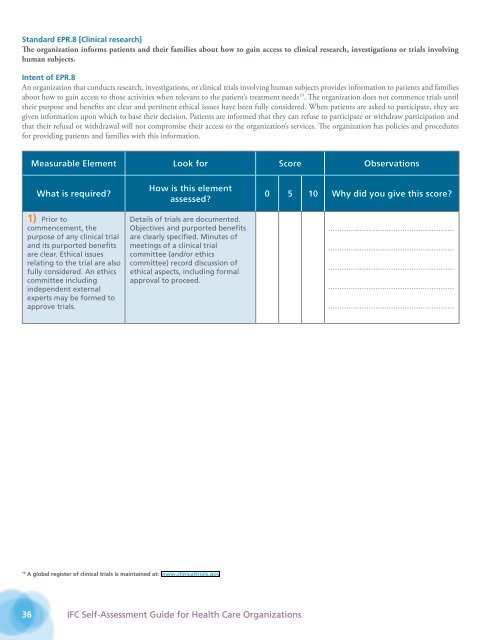
Standard EPR.8 [Clinical research]<br />
The organization in<strong>for</strong>ms patients and their families about how to gain access to clinical research, investigations or trials involving<br />
human subjects.<br />
Intent of EPR.8<br />
An organization that conducts research, investigations, or clinical trials involving human subjects provides in<strong>for</strong>mation to patients and families<br />
about how to gain access to those activities when relevant to the patient’s treatment needs 19 . The organization does not commence trials until<br />
their purpose and benefits are clear and pertinent ethical issues have been fully considered. When patients are asked to participate, they are<br />
given in<strong>for</strong>mation upon which to base their decision. Patients are in<strong>for</strong>med that they can refuse to participate or withdraw participation and<br />
that their refusal or withdrawal will not compromise their access to the organization’s services. The organization has policies and procedures<br />
<strong>for</strong> providing patients and families with this in<strong>for</strong>mation.<br />
Measurable Element Look <strong>for</strong> Score Observations<br />
What is required<br />
1) Prior to<br />
commencement, the<br />
purpose of any clinical trial<br />
and its purported benefits<br />
are clear. Ethical issues<br />
relating to the trial are also<br />
fully considered. An ethics<br />
committee including<br />
independent external<br />
experts may be <strong>for</strong>med to<br />
approve trials.<br />
How is this element<br />
assessed<br />
Details of trials are documented.<br />
Objectives and purported benefits<br />
are clearly specified. Minutes of<br />
meetings of a clinical trial<br />
committee (and/or ethics<br />
committee) record discussion of<br />
ethical aspects, including <strong>for</strong>mal<br />
approval to proceed.<br />
0 5 10 Why did you give this score<br />
........................................................<br />
........................................................<br />
........................................................<br />
........................................................<br />
........................................................<br />
19<br />
A global register of clinical trials is maintained at: www.clinicaltrials.gov<br />
36 <strong>IFC</strong> <strong>Self</strong>-<strong>Assessment</strong> <strong>Guide</strong> <strong>for</strong> <strong>Health</strong> <strong>Care</strong> <strong>Organizations</strong>













![Print a two-page fact sheet on this project [PDF] - IFC](https://img.yumpu.com/43449799/1/190x245/print-a-two-page-fact-sheet-on-this-project-pdf-ifc.jpg?quality=85)


