Staff Members of the Institute of Biochemistry, TU Graz http://www ...
Staff Members of the Institute of Biochemistry, TU Graz http://www ...
Staff Members of the Institute of Biochemistry, TU Graz http://www ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
The absence <strong>of</strong> vitamin B 6 biosyn<strong>the</strong>sis in humans clearly suggests that <strong>the</strong> proteins<br />
involved are interesting new drug targets for <strong>the</strong> development <strong>of</strong> antibiotics, fungicides<br />
and herbicides. The recent discovery <strong>of</strong> vitamin B 6 biosyn<strong>the</strong>sis in <strong>the</strong> causative agent <strong>of</strong><br />
malaria, Plasmodium falciparum, prompted us to engage in an international<br />
collaboration in order to characterise <strong>the</strong> chemistry and enzymology <strong>of</strong> vitamin B 6<br />
biosyn<strong>the</strong>sis in this highly pathogenic organism. Our studies will provide a detailed<br />
understanding <strong>of</strong> <strong>the</strong> role(s) <strong>of</strong> Pdx1 and Pdx2 in P. falciparum and will enable us to<br />
initiate a drug design/screening programme which will receive additional support from<br />
<strong>the</strong> elucidation <strong>of</strong> <strong>the</strong> three-dimensional structures <strong>of</strong> <strong>the</strong> proteins and <strong>the</strong>ir active<br />
complex (<strong>the</strong>sis project <strong>of</strong> Karlheinz Flicker).<br />
NAD(P)H:FMN quinone reductases<br />
Following our mechanistic and structural studies on YcnD, a nitroreductase from<br />
Bacillus subtilis, we have cloned ano<strong>the</strong>r NADPH-dependent flavin containing<br />
oxidoreductase from this organism (termed YhdA). We have achieved high level<br />
expression in E. coli BL 21 host cells and purified it to homogeneity. Steady-state<br />
kinetic studies have shown that YhdA reduces FMN and nitro-organic compounds at <strong>the</strong><br />
expense <strong>of</strong> NADPH as <strong>the</strong> preferred electron donor. In parallel to <strong>the</strong> bacterial enzyme,<br />
we have also started to investigate <strong>the</strong> properties <strong>of</strong> a homologous enzyme from<br />
Saccharomyces cerevisiae (termed LOT6p). Despite <strong>the</strong> availability <strong>of</strong> a threedimensional<br />
x-ray structure for YhdA (1NNI) and LOT6p (1T0I), <strong>the</strong> physiological role<br />
<strong>of</strong> <strong>the</strong> enzyme was unclear. Our recent studies have now demonstrated that both<br />
enzymes rapidly reduce quinones at <strong>the</strong> expense <strong>of</strong> a reduced nicotinamide c<strong>of</strong>actor.<br />
These studies were complemented by localization studies <strong>of</strong> LOT6p in yeast, which<br />
confirmed <strong>the</strong> cytosolic occurrence <strong>of</strong> <strong>the</strong> protein (in collaboration with Pr<strong>of</strong>. Daum, <strong>TU</strong><br />
<strong>Graz</strong>). In order to fur<strong>the</strong>r characterize <strong>the</strong> cellular role <strong>of</strong> LOT6p, we are now in <strong>the</strong><br />
process to search for protein interaction partners (<strong>the</strong>sis project <strong>of</strong> Sonja Sollner and<br />
diploma project <strong>of</strong> Markus Schober).<br />
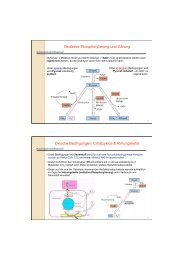
Interestingly, <strong>the</strong> bacterial homolog <strong>of</strong> LOT6p, YhdA, forms stable tetramers instead <strong>of</strong><br />
dimers. Apparently, this higher oligomeric state gives rise to increased <strong>the</strong>rmostability<br />
(Deller et al., 2006). Inspection <strong>of</strong> <strong>the</strong> three-dimensional structure led to <strong>the</strong><br />
identification <strong>of</strong> four salt-bridges, which hold two dimers toge<strong>the</strong>r (see figure).<br />
11