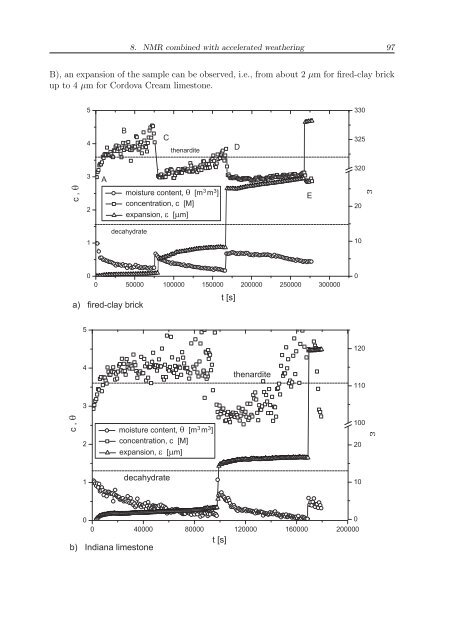
8. NMR combined with accelerated weathering 97 B), an expansion of the sample can be observed, i.e., from about 2 µm for fired-clay brick up to 4 µm for Cordova Cream limestone. c , moisture content, [m3m 3] concentration, c [M] expansion, [ m] a) fired-clay brick t [s] c , moisture content, [m3m 3] concentration, c [M] expansion, [ m] b) Indiana limestone t [s]
8. NMR combined with accelerated weathering 98 c , moisture content, [m3m 3] concentration, c [M] expansion, [ m] c) Cordova cream limestone t [s] Fig. 8.4: Measured moisture content, concentration, and expansion as a function of time for a) fired-clay brick, b) Cordova cream limestone, and c) Indiana limestone. Initially all sample are saturated with a 3 M sodium sulfate solution at 40 ◦ C, after which the samples are cooled down to 22 ◦ C and the experiment is started. In order to obtain more information on the salt transport during drying, we have plotted the same data in a so-called efflorescence pathway diagram (EPD) in figure 8.5 [68]. In an EPD, the total amount of sodium sulfate present in the solution, cθ (in mole/kg), is plotted against the moisture content θ. In such a diagram two limiting situations can be distinguished. First, very slow drying, in which case salt transport by diffusion dominates, i.e., P e ≪ 1. In this case, the ion profiles remain homogeneous and for some time no crystallization will occur. The average sodium sulfate concentration will slowly increase until the complete sample has reached the solubility of thenardite (3.4 M). From this point on any additional drying will result in crystallization. Second, very fast drying, i.e., P e ≫ 1. In this case, ions are directly advected with the moisture to the top of the sample, where a concentration peak with a very small width will develop. If the rate of crystallization is high enough, i.e., if there are enough nucleation sites at the top to form crystals, the average Na 2 SO 4 concentration in the solution in the sample itself will remain almost constant at nearly at the initial concentration. Figure 8.5 shows that for all these experiments P e ≪ 1 during the initial drying in the first cycle, indicating that diffusion is dominant. In this case we expect a homogeneous crystallization behavior throughout the sample and the expansion will be representative for the crystallization in the material. We like to note that these EPDs confirm that thenardite is being formed in the samples. When the relative moisture content in the samples has dropped to about 25 %, the