Section 1.1 Section 1.2 Section 1.3 - The Student Room
Section 1.1 Section 1.2 Section 1.3 - The Student Room
Section 1.1 Section 1.2 Section 1.3 - The Student Room
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
SECTION 13<br />
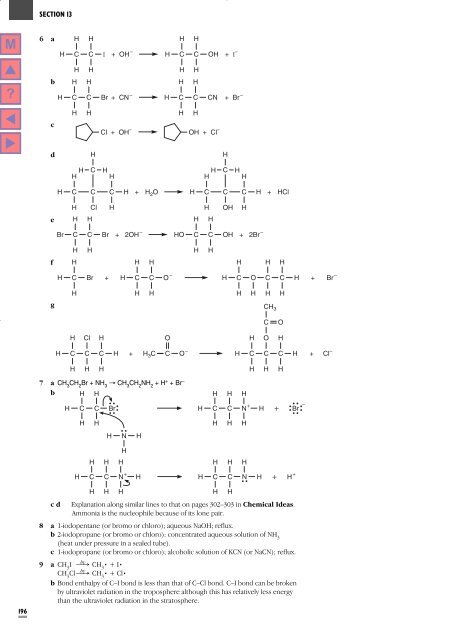
6 a<br />
H<br />
H<br />
H<br />
H<br />
H<br />
C<br />
C<br />
I<br />
+ OH – H C C<br />
OH<br />
+ I –<br />
H<br />
H<br />
H<br />
H<br />
b<br />
H<br />
H<br />
H<br />
H<br />
H<br />
C<br />
C<br />
Br<br />
+ CN – H C C<br />
CN<br />
+ Br –<br />
H<br />
H<br />
H<br />
H<br />
c<br />
Cl + OH – OH + Cl –<br />
d<br />
H<br />
H<br />
H C H H C<br />
H H<br />
H<br />
H<br />
H<br />
H<br />
C<br />
C<br />
C<br />
H<br />
+ H 2 O H C C C H + HCl<br />
H<br />
Cl<br />
H<br />
H<br />
OH<br />
H<br />
e<br />
H<br />
H<br />
H<br />
H<br />
Br<br />
C<br />
C<br />
Br<br />
+ 2OH – HO C C<br />
OH<br />
+ 2Br –<br />
H<br />
H<br />
H<br />
H<br />
f<br />
H<br />
H<br />
H<br />
H<br />
H<br />
H<br />
H<br />
C<br />
Br<br />
+ H C C O –<br />
H C O C C H + Br –<br />
g<br />
H<br />
H<br />
H<br />
H<br />
H<br />
H<br />
CH 3<br />
H<br />
C<br />
O<br />
H<br />
Cl<br />
H<br />
O<br />
H<br />
O<br />
H<br />
H<br />
C<br />
C<br />
C<br />
H<br />
+ H C O –<br />
H C C C H + Cl –<br />
3 C<br />
H<br />
H<br />
H<br />
H<br />
H<br />
H<br />
7 a CH 3<br />
CH 2<br />
Br + NH 3<br />
Æ CH 3<br />
CH 2<br />
NH 2<br />
+ H + +Br –<br />
b H H<br />
H<br />
H<br />
H<br />
H C C Br<br />
H C C<br />
N +<br />
H +<br />
Br<br />
–<br />
H<br />
H<br />
H<br />
H<br />
H<br />
H N H<br />
H<br />
H<br />
H<br />
H<br />
H<br />
H<br />
H<br />
H C C<br />
N +<br />
H H C C<br />
N<br />
H + H +<br />
196<br />
H<br />
H<br />
H<br />
c d Explanation along similar lines to that on pages 302–303 in Chemical Ideas.<br />
Ammonia is the nucleophile because of its lone pair.<br />
8 a 1-iodopentane (or bromo or chloro); aqueous NaOH; reflux.<br />
b 2-iodopropane (or bromo or chloro): concentrated aqueous solution of NH 3<br />
(heat under pressure in a sealed tube).<br />
c 1-iodopropane (or bromo or chloro); alcoholic solution of KCN (or NaCN); reflux.<br />
hv<br />
9 a CH 3<br />
I æÆ CH 3 + I .<br />
hv<br />
CH 3<br />
Cl æÆ CH 3<br />
. + Cl .<br />
b Bond enthalpy of C–I bond is less than that of C–Cl bond. C–I bond can be broken<br />
by ultraviolet radiation in the troposphere although this has relatively less energy<br />
than the ultraviolet radiation in the stratosphere.<br />
H<br />
H















![ISI Web of Knowledge [v.4.10] - All Databases Results - Benjamin-Mills](https://img.yumpu.com/39253071/1/184x260/isi-web-of-knowledge-v410-all-databases-results-benjamin-mills.jpg?quality=85)