Laboratory methods• Diagnostic Laboratory methods that currently are be<strong>in</strong>g used are:• 1-Rapid methods of presumptive diagnosis ,with two approach available, a) Prepar<strong>in</strong>g Wet mounts,Dem<strong>on</strong>strate the hypertrophied nuclei <strong>in</strong> squash preparati<strong>on</strong>s of the gills and/or cuticular epithelium, whichcan be sta<strong>in</strong>ed or unsta<strong>in</strong>ed, b)Prepar<strong>in</strong>g Smears, Dem<strong>on</strong>strate WSSV aggregates <strong>in</strong> unsta<strong>in</strong>ed smearpreparati<strong>on</strong>s of haemolymph by dark-field microscopy. This is the simplest of the microscopic techniques andis recommended for people with limited expertise <strong>in</strong> WSSV. The aggregates appear as small reflective spots of0.5 μm <strong>in</strong> diameter .• 2- Histopathology,with prepar<strong>in</strong>g fixed secti<strong>on</strong>s that dem<strong>on</strong>strates pathognom<strong>on</strong>ic <strong>in</strong>clusi<strong>on</strong> bodies <strong>in</strong> targettissues.The secti<strong>on</strong>s must be exam<strong>in</strong>ed by light microscopy for the presence of moderate to large numbers ofhypertrophied nuclei with eos<strong>in</strong>ophilic to basophilic central <strong>in</strong>clusi<strong>on</strong>s surrounded by marg<strong>in</strong>ated chromat<strong>in</strong> <strong>in</strong>tissues of ectodermal and mesodermal orig<strong>in</strong>. The best tissues for exam<strong>in</strong>ati<strong>on</strong> are the subcuticular tissues ofthe stomach, cephalothorax or gill .• 3- In situ DNA hybridizati<strong>on</strong>, by us<strong>in</strong>g WSSV-specific DNA probes with histological secti<strong>on</strong>s to dem<strong>on</strong>strate thepresence of WSSV nuclei acid <strong>in</strong> <strong>in</strong>fected cells.• 4-Bioassay method, that will c<strong>on</strong>firm the presence of a pathogenic virus, but does not identify the specificvirus. Therefore, bioassay must be used <strong>in</strong> c<strong>on</strong>juncti<strong>on</strong> with laboratory tests to c<strong>on</strong>firm the identity of thevirus. If SPF shrimp are available, the bioassay method is based <strong>on</strong> Nunan L.M.et al. (1998) and Durand S.V. etal. (2000), is suitable for WSSV diagnosis recommended by <strong>OIE</strong> aquatic manual.In situ DNA hybridizati<strong>on</strong>,Source: D.V.lightner• 5- Transmissi<strong>on</strong> Electr<strong>on</strong> Microscopy(TEM). Dem<strong>on</strong>strates the virus <strong>in</strong> tissue secti<strong>on</strong>s or <strong>in</strong> semi-purifiednegatively sta<strong>in</strong>ed virus preparati<strong>on</strong>s (e.g.from haemolymph).• 6- Antibody-based assays,There are various immunological assays <strong>in</strong>clud<strong>in</strong>g western blot analysis, immunodotassay, <strong>in</strong>direct fluorescent antibody test (IFAT), immunohistochemistry (IHC) or enzyme l<strong>in</strong>ked immunosorbentassay (ELISA) to detect WSSV by us<strong>in</strong>g both polycl<strong>on</strong>al and m<strong>on</strong>ocl<strong>on</strong>al antibodies raised aga<strong>in</strong>st either thevirus or a recomb<strong>in</strong>ant viral structural prote<strong>in</strong>. Antibody-based methods can be fast, c<strong>on</strong>venient andapplicable to field use, but as they have <strong>on</strong>ly about the same sensitivity as 1-step PCR, they are recommended<strong>on</strong>ly to c<strong>on</strong>firm acute WSD.• 7- Polymerase Cha<strong>in</strong> Reacti<strong>on</strong> test (PCR) ,Two –step PCR test is the preferred recommended test by <strong>OIE</strong> aquaticmanual. The protocol described <strong>in</strong> <strong>OIE</strong> aquatic manual is from Lo C.F. et al. (1997), and is recommended for allsituati<strong>on</strong>s where WSSV diagnosis is required. A positive result <strong>in</strong> the first step of this standard protocol impliesa serious WSSV <strong>in</strong>fecti<strong>on</strong>, whereas, when a positive result is obta<strong>in</strong>ed <strong>in</strong> the sec<strong>on</strong>d amplificati<strong>on</strong> step <strong>on</strong>ly, alatent or carrier-state <strong>in</strong>fecti<strong>on</strong> is <strong>in</strong>dicated. Alternative assays have also been described, but are notrecommended unless they have first been compared with the protocol described there.• Many countries have developed the PCR test by design<strong>in</strong>g primers from the virus stra<strong>in</strong> <strong>in</strong> their country <strong>in</strong>order to prepare a more specific diagnostic kits for detecti<strong>on</strong> of WSSV. Iranians as well, have designed such kitby us<strong>in</strong>g a designated primer orig<strong>in</strong>ally from VP24 prote<strong>in</strong> of the virus <strong>in</strong> the diseased shrimp.•• PCR commercial kits are available for WSSV diagnosis and are acceptable provided they have been validated asfit for such purpose. Please c<strong>on</strong>sult the <strong>OIE</strong> Register for kits that have been certified by the <strong>OIE</strong>(http://www.oie.<strong>in</strong>t/vcda/eng/en_vcda_registre.htm).• 8- DNA Sequenc<strong>in</strong>g of PCR products, For c<strong>on</strong>firmati<strong>on</strong> of suspected new hosts of WSSV, the DNA fragmentamplified from the two-step nested diagnostic PCR should be sequenced. The cl<strong>on</strong><strong>in</strong>g and sequenc<strong>in</strong>gprotocols described <strong>in</strong> <strong>OIE</strong> aquatic manual are accord<strong>in</strong>g to Clayd<strong>on</strong> K. et al. (2004).Picture: M. Afsharnasab, Outbreak <strong>in</strong> Khuzestan prov<strong>in</strong>ce, Iran, 2008.The designated primer, orig<strong>in</strong>ally by VP24 with 414 bp from WSSV is used <strong>in</strong>the PCR test.
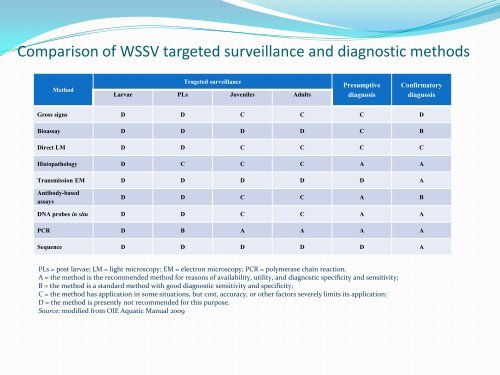
Comparis<strong>on</strong> of WSSV targeted surveillance and diagnostic methodsMethodTrageted surveillanceLarvae PLs Juveniles AdultsPresumptivediagnosisC<strong>on</strong>firmatorydiagnosisGross signs D D C C C DBioassay D D D D C BDirect LM D D C C C CHistopathology D C C C A ATransmissi<strong>on</strong> EM D D D D D AAntibody-basedassaysD D C C A BDNA probes <strong>in</strong> situ D D C C A APCR D B A A A ASequence D D D D D APLs = post larvae; LM = light microscopy; EM = electr<strong>on</strong> microscopy; PCR = polymerase cha<strong>in</strong> reacti<strong>on</strong>.A = the method is the recommended method for reas<strong>on</strong>s of availability, utility, and diagnostic specificity and sensitivity;B = the method is a standard method with good diagnostic sensitivity and specificity;C = the method has applicati<strong>on</strong> <strong>in</strong> some situati<strong>on</strong>s, but cost, accuracy, or other factors severely limits its applicati<strong>on</strong>;D = the method is presently not recommended for this purpose.Source: modified from <strong>OIE</strong> Aquatic Manual 2009