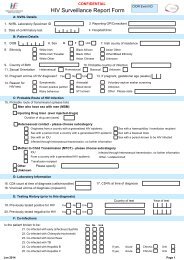
<str<strong>on</strong>g>Guidel<strong>in</strong>es</str<strong>on</strong>g> <strong>on</strong> <strong>the</strong> Preventi<strong>on</strong> <strong>and</strong> C<strong>on</strong>trol <strong>of</strong> <strong>Tuberculosis</strong> <strong>in</strong> Irel<strong>and</strong> 2010HSE/HPSCc<strong>on</strong>ta<strong>in</strong>ment.Although <strong>the</strong> Seventh Schedule recommends <strong>the</strong> follow<strong>in</strong>g po<strong>in</strong>ts, <strong>the</strong> Nati<strong>on</strong>al TB Advisory Committee ispropos<strong>in</strong>g that <strong>the</strong>se recommendati<strong>on</strong>s outl<strong>in</strong>ed below should be c<strong>on</strong>sidered compulsory.• The workplace is to be separated from any o<strong>the</strong>r activities <strong>in</strong> <strong>the</strong> same build<strong>in</strong>g• The workplace is to be sealable to permit dis<strong>in</strong>fecti<strong>on</strong>• The workplace is to be ma<strong>in</strong>ta<strong>in</strong>ed at an air pressure negative to atmosphere• An observati<strong>on</strong> w<strong>in</strong>dow, or an alternative, is to be present, so that occupants can be seen <strong>and</strong>• A laboratory is to c<strong>on</strong>ta<strong>in</strong> its own equipment.The decisi<strong>on</strong> to make this proposal has been <strong>in</strong>fluenced by look<strong>in</strong>g at best <strong>in</strong>ternati<strong>on</strong>al practice <strong>in</strong>relati<strong>on</strong> to <strong>the</strong> items that are recommended. In <strong>the</strong> UK, <strong>the</strong> C<strong>on</strong>trol <strong>of</strong> Substances Hazardous to Healthregulati<strong>on</strong>s 1994 (COSHH) implements <strong>the</strong> EC Biological Agents Directive. The Advisory Committee <strong>on</strong>Dangerous Pathogens (UK) (ACDP) orig<strong>in</strong>ally published <strong>the</strong> Categorisati<strong>on</strong> <strong>of</strong> biological agents accord<strong>in</strong>gto hazard <strong>and</strong> categories <strong>of</strong> c<strong>on</strong>ta<strong>in</strong>ment (4 th editi<strong>on</strong>) 1995. 199 This guidance has now been replaced by Themanagement, design <strong>and</strong> operati<strong>on</strong> <strong>of</strong> microbiological c<strong>on</strong>ta<strong>in</strong>ment laboratories, 2001. 200 This publicati<strong>on</strong>complements <strong>the</strong> Health <strong>and</strong> Safety Commissi<strong>on</strong>’s Health Service Advisory Committee’s (HSAC) guidance<strong>on</strong> Safe work<strong>in</strong>g <strong>and</strong> <strong>the</strong> preventi<strong>on</strong> <strong>of</strong> <strong>in</strong>fecti<strong>on</strong> <strong>in</strong> cl<strong>in</strong>ical laboratories <strong>and</strong> similar facilities 201 <strong>and</strong> <strong>the</strong> ACDPpublicati<strong>on</strong> Biological agents: Manag<strong>in</strong>g <strong>the</strong> risks <strong>in</strong> laboratories <strong>and</strong> healthcare premises. 202 Under thisguidance <strong>the</strong> above recommendati<strong>on</strong>s are compulsory.In <strong>the</strong> USA, laboratories perform<strong>in</strong>g functi<strong>on</strong>s at ATS level 2 or 3 must use Biosafety Level 3. Biosafety level3 is very similar to C<strong>on</strong>ta<strong>in</strong>ment Level 3 (CL3) <strong>and</strong> <strong>the</strong> above recommendati<strong>on</strong>s are <strong>in</strong>cluded as rules. 114;203F<strong>in</strong>ally, a CL3 Laboratory will fail to achieve accreditati<strong>on</strong> with Cl<strong>in</strong>ical Pathology Accreditati<strong>on</strong> (UK) Ltd if<strong>the</strong>se recommendati<strong>on</strong>s are not <strong>in</strong> place.Recommendati<strong>on</strong>:Recommendati<strong>on</strong>s <strong>in</strong> <strong>the</strong> Seventh Schedule <strong>of</strong> <strong>the</strong> S.I. No. 146/1994 Safety, Health <strong>and</strong>Welfare at Work (Biological Agents) Regulati<strong>on</strong>s 1994 should be <strong>in</strong>terpreted as m<strong>and</strong>atory <strong>in</strong>relati<strong>on</strong> to work<strong>in</strong>g with M. tuberculosis complex.Transportati<strong>on</strong>The transport <strong>of</strong> <strong>in</strong>fectious substances by road, rail, sea <strong>and</strong> air are each <strong>the</strong> subject <strong>of</strong> <strong>in</strong>ternati<strong>on</strong>alregulati<strong>on</strong>, whose whole descripti<strong>on</strong> is bey<strong>on</strong>d <strong>the</strong> remit <strong>of</strong> <strong>the</strong>se guidel<strong>in</strong>es. The relevant legislati<strong>on</strong> <strong>in</strong>Irel<strong>and</strong> for transport by road is <strong>the</strong> “Carriage <strong>of</strong> Dangerous Goods by Road” Regulati<strong>on</strong>s 2007 StatutoryInstrument No. 288 <strong>of</strong> 2007 204 <strong>and</strong> <strong>the</strong> 2007 ADR regulati<strong>on</strong>s. 205SpecimensBiological substances that have been correctly classified as UN No. 3373 which are packaged <strong>and</strong> marked<strong>in</strong> accordance with packag<strong>in</strong>g <strong>in</strong>structi<strong>on</strong> P650, are not subject to any o<strong>the</strong>r requirements <strong>of</strong> <strong>the</strong> 2007 ADRregulati<strong>on</strong>s. This implies that provided <strong>the</strong> correct packag<strong>in</strong>g is used, <strong>the</strong>re is no requirement for any ADRdocumentati<strong>on</strong> or for <strong>the</strong> requirement to have specially marked <strong>and</strong> equipped vehicles or tra<strong>in</strong>ed drivers.In summary, any method <strong>of</strong> transportati<strong>on</strong> can be used to transport patient specimens by road <strong>on</strong>ceproperly packaged <strong>and</strong> labelled.CulturesFor transport purposes, pathogens are assigned to two categories, A <strong>and</strong> B. Category A <strong>in</strong>cludes <strong>the</strong>higher risk <strong>in</strong>fectious microorganisms such as M. tuberculosis complex but <strong>on</strong>ly when it is present as aculture. Never<strong>the</strong>less, when cultures that are be<strong>in</strong>g transported by road are <strong>in</strong>tended for diagnostic orcl<strong>in</strong>ical purposes (not research), <strong>the</strong>y may be classified as <strong>in</strong>fectious substances <strong>of</strong> Category B. Infectioussubstances <strong>in</strong> Category B shall be assigned to UN No. 3373 <strong>and</strong> <strong>the</strong> proper shipp<strong>in</strong>g name <strong>of</strong> UN No. 3373-57-
<str<strong>on</strong>g>Guidel<strong>in</strong>es</str<strong>on</strong>g> <strong>on</strong> <strong>the</strong> Preventi<strong>on</strong> <strong>and</strong> C<strong>on</strong>trol <strong>of</strong> <strong>Tuberculosis</strong> <strong>in</strong> Irel<strong>and</strong> 2010HSE/HPSCis “BIOLOGICAL SUBSTANCE, CATEGORY B”. These are transported <strong>in</strong> accordance with ADR packag<strong>in</strong>gregulati<strong>on</strong>s P650.However, at <strong>the</strong> time <strong>of</strong> publicati<strong>on</strong> <strong>of</strong> this document, when cultures <strong>of</strong> M. tuberculosis complex arebe<strong>in</strong>g transported by air or sea <strong>the</strong>y must follow <strong>the</strong> Internati<strong>on</strong>al Air Transport Associati<strong>on</strong> (IATA) <strong>and</strong><strong>the</strong> Internati<strong>on</strong>al Maritime Dangerous Goods (IMDG) regulati<strong>on</strong>s, that currently assign M. tuberculosisto Category A <strong>and</strong> <strong>the</strong>y must be transported as UN No. 2814 “INFECTIOUS SUBSTANCE AFFECTINGHUMANS” <strong>and</strong> packaged <strong>in</strong> accordance with pack<strong>in</strong>g <strong>in</strong>structi<strong>on</strong> P620. 205 This is most easily achieved byus<strong>in</strong>g an approved <strong>and</strong> licensed courier.M. tuberculosis complex outside <strong>the</strong> CL3 laboratoryIt is worth not<strong>in</strong>g that M. tuberculosis has been recovered from many body sites. 206-209 It follows <strong>the</strong>reforethat appropriate risk assessments with regard to M. tuberculosis are carried out when process<strong>in</strong>g samplesfor o<strong>the</strong>r pathogens <strong>in</strong> o<strong>the</strong>r parts <strong>of</strong> <strong>the</strong> laboratory. This is especially so when an extended <strong>in</strong>cubati<strong>on</strong> timeis applied to recover fastidious pathogens. The observati<strong>on</strong> that M. tuberculosis can be grown with<strong>in</strong> sixdays <strong>on</strong> blood agar <strong>and</strong> that blood agar is at least as efficient as Lowenste<strong>in</strong> Jensen medium <strong>in</strong> recover<strong>in</strong>gM. tuberculosis from respiratory <strong>and</strong> lymph node aspirates must be c<strong>on</strong>sidered when perform<strong>in</strong>g riskassessment analysis for o<strong>the</strong>r procedures <strong>in</strong> <strong>the</strong> microbiology laboratory. 210 Respiratory specimens, <strong>in</strong>particular, may c<strong>on</strong>ta<strong>in</strong> viable M. tuberculosis organisms <strong>and</strong> <strong>the</strong>re are recommendati<strong>on</strong>s that <strong>the</strong>se shouldbe analysed <strong>in</strong> a CL3 facility. 199;211Recommendati<strong>on</strong>:All relevant legislati<strong>on</strong> (nati<strong>on</strong>al <strong>and</strong> <strong>in</strong>ternati<strong>on</strong>al) for <strong>the</strong> transport <strong>and</strong> h<strong>and</strong>l<strong>in</strong>g <strong>of</strong> specimens<strong>and</strong> cultures for tuberculosis should be strictly adhered to at all times.Molecular test<strong>in</strong>gTests <strong>in</strong>volv<strong>in</strong>g molecular methods do not require CL3 practices after <strong>the</strong> organism has been rendered n<strong>on</strong>viable.This is normally achieved through <strong>the</strong> applicati<strong>on</strong> <strong>of</strong> heat. Some systems have been shown not torender <strong>the</strong> cultures completely unviable <strong>and</strong> <strong>the</strong>refore a laboratory must be able to def<strong>in</strong>itively state <strong>and</strong>dem<strong>on</strong>strate over time that any protocol <strong>in</strong> use for extract<strong>in</strong>g mycobacterial DNA/RNA has succeeded <strong>in</strong>render<strong>in</strong>g <strong>the</strong> sample n<strong>on</strong>-<strong>in</strong>fectious before its removal from a CL3 facility. Methods which have producedc<strong>on</strong>sistent kill<strong>in</strong>g <strong>of</strong> all mycobacterial species tested are those by which <strong>the</strong> tubes were fully immersed <strong>in</strong>boil<strong>in</strong>g water or with<strong>in</strong> a forced-hot-air oven set at 100 o C. 212-214AuditThere should be regular safety audits <strong>of</strong> <strong>the</strong> CL3 premises <strong>and</strong> processes.4.8 Quality AssuranceModern laboratory practice necessitates participati<strong>on</strong> <strong>in</strong> both an <strong>in</strong>ternal <strong>and</strong> an approved external qualityassurance scheme e.g. United K<strong>in</strong>gdom Nati<strong>on</strong>al External Quality Assessment Service (NEQAS). Qualityassurance is a system that m<strong>on</strong>itors <strong>and</strong> improves <strong>the</strong> efficiency <strong>and</strong> reliability <strong>of</strong> <strong>the</strong> laboratory service bypay<strong>in</strong>g attenti<strong>on</strong> to detail at every step. There are three phases to <strong>the</strong> process as follows:Pre-analytical activities• How <strong>the</strong> test is ordered• Specimen collecti<strong>on</strong> procedures• Transport to <strong>the</strong> laboratory• Specimen h<strong>and</strong>l<strong>in</strong>g <strong>and</strong> storage <strong>and</strong>• Completeness <strong>of</strong> patient <strong>in</strong>formati<strong>on</strong>.-58-