2005 Edition Report on Drug Administration Procedure & Practices ...
2005 Edition Report on Drug Administration Procedure & Practices ...
2005 Edition Report on Drug Administration Procedure & Practices ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
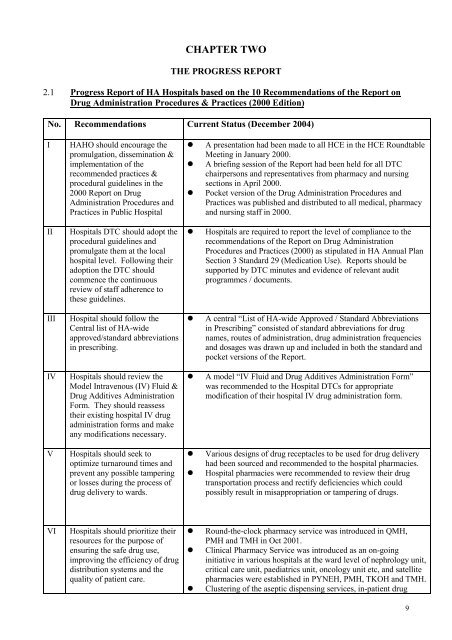
CHAPTER TWOTHE PROGRESS REPORT2.1 Progress <str<strong>on</strong>g>Report</str<strong>on</strong>g> of HA Hospitals based <strong>on</strong> the 10 Recommendati<strong>on</strong>s of the <str<strong>on</strong>g>Report</str<strong>on</strong>g> <strong>on</strong><strong>Drug</strong> Administrati<strong>on</strong> <strong>Procedure</strong>s & <strong>Practices</strong> (2000 <str<strong>on</strong>g>Editi<strong>on</strong></str<strong>on</strong>g>)No. Recommendati<strong>on</strong>s Current Status (December 2004)IIIIIIIVVHAHO should encourage thepromulgati<strong>on</strong>, disseminati<strong>on</strong> &implementati<strong>on</strong> of therecommended practices &procedural guidelines in the2000 <str<strong>on</strong>g>Report</str<strong>on</strong>g> <strong>on</strong> <strong>Drug</strong>Administrati<strong>on</strong> <strong>Procedure</strong>s and<strong>Practices</strong> in Public HospitalHospitals DTC should adopt theprocedural guidelines andpromulgate them at the localhospital level. Following theiradopti<strong>on</strong> the DTC shouldcommence the c<strong>on</strong>tinuousreview of staff adherence tothese guidelines.Hospital should follow theCentral list of HA-wideapproved/standard abbreviati<strong>on</strong>sin prescribing.Hospitals should review theModel Intravenous (IV) Fluid &<strong>Drug</strong> Additives Administrati<strong>on</strong>Form. They should reassesstheir existing hospital IV drugadministrati<strong>on</strong> forms and makeany modificati<strong>on</strong>s necessary.Hospitals should seek tooptimize turnaround times andprevent any possible tamperingor losses during the process ofdrug delivery to wards.• A presentati<strong>on</strong> had been made to all HCE in the HCE RoundtableMeeting in January 2000.• A briefing sessi<strong>on</strong> of the <str<strong>on</strong>g>Report</str<strong>on</strong>g> had been held for all DTCchairpers<strong>on</strong>s and representatives from pharmacy and nursingsecti<strong>on</strong>s in April 2000.• Pocket versi<strong>on</strong> of the <strong>Drug</strong> Administrati<strong>on</strong> <strong>Procedure</strong>s and<strong>Practices</strong> was published and distributed to all medical, pharmacyand nursing staff in 2000.• Hospitals are required to report the level of compliance to therecommendati<strong>on</strong>s of the <str<strong>on</strong>g>Report</str<strong>on</strong>g> <strong>on</strong> <strong>Drug</strong> Administrati<strong>on</strong><strong>Procedure</strong>s and <strong>Practices</strong> (2000) as stipulated in HA Annual PlanSecti<strong>on</strong> 3 Standard 29 (Medicati<strong>on</strong> Use). <str<strong>on</strong>g>Report</str<strong>on</strong>g>s should besupported by DTC minutes and evidence of relevant auditprogrammes / documents.• A central “List of HA-wide Approved / Standard Abbreviati<strong>on</strong>sin Prescribing” c<strong>on</strong>sisted of standard abbreviati<strong>on</strong>s for drugnames, routes of administrati<strong>on</strong>, drug administrati<strong>on</strong> frequenciesand dosages was drawn up and included in both the standard andpocket versi<strong>on</strong>s of the <str<strong>on</strong>g>Report</str<strong>on</strong>g>.• A model “IV Fluid and <strong>Drug</strong> Additives Administrati<strong>on</strong> Form”was recommended to the Hospital DTCs for appropriatemodificati<strong>on</strong> of their hospital IV drug administrati<strong>on</strong> form.• Various designs of drug receptacles to be used for drug deliveryhad been sourced and recommended to the hospital pharmacies.• Hospital pharmacies were recommended to review their drugtransportati<strong>on</strong> process and rectify deficiencies which couldpossibly result in misappropriati<strong>on</strong> or tampering of drugs.VIHospitals should prioritize theirresources for the purpose ofensuring the safe drug use,improving the efficiency of drugdistributi<strong>on</strong> systems and thequality of patient care.• Round-the-clock pharmacy service was introduced in QMH,PMH and TMH in Oct 2001.• Clinical Pharmacy Service was introduced as an <strong>on</strong>-goinginitiative in various hospitals at the ward level of nephrology unit,critical care unit, paediatrics unit, <strong>on</strong>cology unit etc, and satellitepharmacies were established in PYNEH, PMH, TKOH and TMH.• Clustering of the aseptic dispensing services, in-patient drug9