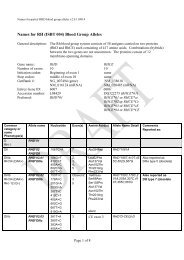
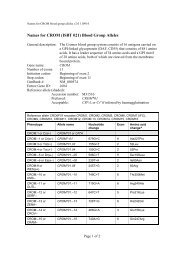
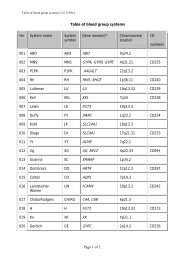
10 Guidel<strong>in</strong>edoes not supply <strong>RFID</strong> tags as part <strong>of</strong> <strong>the</strong> donation set. Insome organizations, it may not be possible to providepatient identity on <strong>the</strong> tag because <strong>of</strong> data security legislation.As each ISBT 128 data structure is uniquely identifiable[1], it may not be necessary to def<strong>in</strong>e an overall fixedtag memory structure but it would probably be more efficient,with respect to IT systems, if a standard structurewere <strong>in</strong> place.Tag Manufacturers<strong>RFID</strong> tag manufacturers encode <strong>the</strong> UID, which is designedto be unique globally, on <strong>the</strong> chip. Part <strong>of</strong> this code is anAFI and <strong>for</strong> transfusion medic<strong>in</strong>e <strong>the</strong> code ‘‘BBH’’ wasrequested from <strong>the</strong> ISO committee. The UID code is unique;<strong>the</strong>re<strong>for</strong>e, it may be <strong>the</strong> only data item on <strong>the</strong> tag that isbe<strong>in</strong>g read dur<strong>in</strong>g rout<strong>in</strong>e process<strong>in</strong>g.Blood Bag ManufacturersISBT128 provides data structures perta<strong>in</strong><strong>in</strong>g to manufacturers<strong>of</strong> blood bags, namely <strong>the</strong> ‘Conta<strong>in</strong>er Manufacturer andCatalogue Number’, <strong>the</strong> ‘Conta<strong>in</strong>er Lot Number’, and <strong>the</strong>‘Expiry Date’. The m<strong>in</strong>imum data elements needed are <strong>the</strong>Catalogue Number and Lot Number [1].It may be possible to go fur<strong>the</strong>r than <strong>the</strong>se standards. Forexample, <strong>the</strong> current lot number relates to a batch <strong>of</strong> manyunits <strong>the</strong>re<strong>for</strong>e, <strong>in</strong> <strong>the</strong> event <strong>of</strong> a recall, <strong>the</strong> number <strong>of</strong> packsimplicated can be high. However, if <strong>the</strong> UID is used <strong>in</strong> <strong>the</strong>manufactur<strong>in</strong>g process, considerable benefits should bega<strong>in</strong>ed <strong>for</strong> all parties as it may be possible to limit <strong>the</strong> scope<strong>of</strong> a recall.Bag manufacturer data should be locked on <strong>the</strong> tag aspart <strong>of</strong> <strong>the</strong> pack manufactur<strong>in</strong>g operation. It should not bepossible to amend this data once written; it should belocked irrevocably.Blood CentersISBT 128 def<strong>in</strong>es data structures <strong>for</strong> use by blood centers[1]. It will be necessary to align <strong>the</strong> data structures with <strong>the</strong>data blocks on <strong>the</strong> tag. Twelve data blocks are reserved <strong>for</strong>use by blood centers. Mandatory data <strong>in</strong>cludes donationnumber, product ID and expiration date ⁄ time, and ABO ⁄ Rh.Donation number should be encoded and locked at <strong>the</strong> earliestopportunity. This may be as soon as <strong>the</strong> donation iscollected and donation number label sets are applied to <strong>the</strong>bag set. Alternatively, this data may be encoded when <strong>the</strong>donor ⁄ donation l<strong>in</strong>k is made on an IT system. O<strong>the</strong>r collectiondata could be encoded and locked at <strong>the</strong> same time.The data blocks reserved <strong>for</strong> product code and expirationdate ⁄ time are rewritable to allow <strong>for</strong> remanufactur<strong>in</strong>g <strong>in</strong><strong>the</strong> same bag, such as irradiation. ABO ⁄ Rh is written onceand locked.Blood center data on <strong>the</strong> <strong>RFID</strong> tag will normally belocked as part <strong>of</strong> <strong>the</strong> shipp<strong>in</strong>g process. However,flexibility will be likely to be needed to allow product<strong>in</strong><strong>for</strong>mation lock<strong>in</strong>g at any time. For example, a bloodcenter could issue a unit <strong>of</strong> red cells subsequently irradiatedby a hospital. In this case, it would be necessary toallow <strong>the</strong> hospital to update <strong>the</strong> product <strong>in</strong><strong>for</strong>mation and,potentially, <strong>the</strong> expiration date. Alternatively, productdata could be locked by <strong>the</strong> blood center. The lock<strong>in</strong>gunder such scenarios is likely to require flexibility at anational level.Because <strong>of</strong> <strong>the</strong> limited tag capacity available, it may benecessary to use a Special Test<strong>in</strong>g data structure related to<strong>the</strong> product be<strong>in</strong>g issued. One <strong>of</strong> <strong>the</strong> limitations <strong>of</strong> currentl<strong>in</strong>ear barcode symbols is that it is difficult to fit specialtest<strong>in</strong>g data with<strong>in</strong> <strong>the</strong> available space. The higher memorycapacity <strong>in</strong> <strong>RFID</strong> tags will provide an alternative solution<strong>for</strong> a long-stand<strong>in</strong>g label space problem.HospitalsHospitals and transfusion services can add data to <strong>the</strong> tagregard<strong>in</strong>g <strong>the</strong> <strong>in</strong>tended recipient. Hemovigilance studiesshow that many serious adverse <strong>in</strong>cidents are because <strong>of</strong>errors that occur at <strong>the</strong> patient <strong>in</strong>terface. This is an areawhere <strong>RFID</strong> can make a significant difference.It must be recognized that application <strong>of</strong> patient data to<strong>the</strong> <strong>RFID</strong>-tag will be subject to national patient privacy anddata security legislation and may not be permissible.Where hospitals can add this <strong>in</strong><strong>for</strong>mation, it will be necessaryto determ<strong>in</strong>e what sort <strong>of</strong> <strong>in</strong><strong>for</strong>mation is required by<strong>the</strong> hospitals <strong>for</strong> positive patient identification. For <strong>the</strong> purposes<strong>of</strong> this guidel<strong>in</strong>e, it has been assumed that 96 byteswould be sufficient.Examples <strong>of</strong> tag structureThe follow<strong>in</strong>g three examples show how product-specificISBT128 data structures could be presented on a tag whileensur<strong>in</strong>g that <strong>the</strong> data is aligned with <strong>the</strong> four-bytelock<strong>in</strong>g blocks (Figs 5–7). With<strong>in</strong> each colored block, <strong>the</strong>symbols use <strong>the</strong> same <strong>for</strong>m as with<strong>in</strong> <strong>the</strong> ISBT 128 specification[1].4.6 DisposalThe destruction <strong>of</strong> passive <strong>RFID</strong> tags can follow standardbiohazard destruction protocols used <strong>for</strong> blood bags (medicalwaste <strong>in</strong>c<strong>in</strong>erators).If active <strong>RFID</strong> tags are used on conta<strong>in</strong>ers, recycl<strong>in</strong>gshould be addressed to con<strong>for</strong>m with exist<strong>in</strong>g recommendations<strong>for</strong> battery recycl<strong>in</strong>g.<strong>RFID</strong> tags do not fall with<strong>in</strong> <strong>the</strong> scope <strong>of</strong> <strong>the</strong> EU directive2002 ⁄ 96 ⁄ EC on Waste Electrical and Electronic Equipment(WEEE) if <strong>the</strong> tag is not attached to o<strong>the</strong>r electronic equipmentwhich falls with<strong>in</strong> <strong>the</strong> scope <strong>of</strong> <strong>the</strong> WEEE directive[19].Ó 2010 The Author(s)Journal compilation Ó 2010 International Society <strong>of</strong> Blood <strong>Transfusion</strong>, Vox Sangu<strong>in</strong>is (2010) 98 (Suppl. 2), 1–24
Guidel<strong>in</strong>e 11Fig. 5 Red Cell Unit. Data Structure 012 is relevant to red cell or whole blood products.Platelet Unit1 2 3 4 5 6 7 8 9 101112131415161718192021222324= ) b q q w w w w w w w & ) x x x x x x x x x x Pack Manufacturers In<strong>for</strong>mation/Extended Audit data? ? ? ? ? ? ? ? = α p p p p y y n n n n n n f f Pack Manufacturers Extended Info/Donation No= % g g r e = < a o o o o t d s & > c y y j Blood Groups/Product Code and Expiration Date/Timej j h h m m & ( z z z z z & { A A A A B B Expiration/Special Test<strong>in</strong>g : General/Platelet HLA and Platelet-Specific AntigensB B C C C C C C C C D DPlatelet HLA and Platelet-Specific Antigens& * c y y j j j h h m m = ? ? ? Collection Date and Time/Fit <strong>for</strong> issue/transfusion marker& # a a | | x x x x x x x x x x x x x x x x x x Patient Identification Number? ? z z z z z z z z z z z z z z z z z z z z z z Patient Name and Genderz z z z z z z z z z z z = # a a y y y y m m d d Patient Date <strong>of</strong> BirthData structure 014 is relevant to platelet products.Fig. 6 Platelet Unit. Data structure 014 is relevant to platelet products.Cellular Therapy Product1 2 3 4 5 6 7 8 9 101112131415161718192021222324= ) b q q w w w w w w w & ) x x x x x x x x x x Pack Manufacturers In<strong>for</strong>mation/Extended Audit data? ? ? ? ? ? ? ? = α p p p p y y n n n n n n f f Pack Manufacturers Extended Info/Donation No= % g g r e = < a o o o o t d s & > c y y j Blood Groups/Product Code and Expiration Date/Timej j h h m m & ( z z z z z = [ E E E E F F Expiration/Special Test<strong>in</strong>g : General/Special Test<strong>in</strong>g: Genomically-Determ<strong>in</strong>ed HLA-F F G G G G H H H H L M = " I I I I J J J J M M Special Test<strong>in</strong>g: Genomically-Determ<strong>in</strong>ed HLA-A and -B AllelesM M M M M M M M & * c y y j j j h h m m = ? ? ? Special Test<strong>in</strong>g: Genomically-Determ<strong>in</strong>ed HLA-A and -B Alleles/Collection Date and& # a a | | x x x x x x x x x x x x x x x x x x Patient Identification Number? ? z z z z z z z z z z z z z z z z z z z z z z Patient Name and Genderz z z z z z z z z z z z = # a a y y y y m m d d Patient Date <strong>of</strong> BirthData structure 015 is relevant to cellular <strong>the</strong>rapy products.Fig. 7 Cellular Therapy Product. Data structure 015 is relevant to cellular <strong>the</strong>rapy products.5. Technical Architecture OverviewThe <strong>RFID</strong>-enabled technology <strong>for</strong> transfusion medic<strong>in</strong>e canbe categorized <strong>in</strong> five high-level layers (doma<strong>in</strong>s):• Bus<strong>in</strong>ess processes• <strong>RFID</strong> devices• <strong>RFID</strong> middleware• Enterprise plat<strong>for</strong>m• Enterprise Application IntegrationThese layers and an overall system representation areshown <strong>in</strong> Fig. 8.The Bus<strong>in</strong>ess Process Doma<strong>in</strong>In <strong>the</strong> real world physical items move and processes areexecuted by both mach<strong>in</strong>es and people. This <strong>the</strong>re<strong>for</strong>e creates<strong>the</strong> demand <strong>for</strong> <strong>in</strong><strong>for</strong>mation systems to support andoptimize processes. Data that is generated and capturedrequires <strong>in</strong><strong>for</strong>mation systems to turn it <strong>in</strong>to useful transactional<strong>in</strong><strong>for</strong>mation <strong>for</strong> <strong>the</strong> execution <strong>of</strong> those processes; it isaggregated <strong>for</strong> analysis <strong>of</strong> trends and process optimization.In order to fully support this doma<strong>in</strong>, additional customs<strong>of</strong>tware components (i.e. database schema, user <strong>in</strong>terface,<strong>in</strong>tegration between process applications) are needed.<strong>RFID</strong> Device Doma<strong>in</strong>The <strong>RFID</strong> Device Doma<strong>in</strong> refers to <strong>the</strong> technical <strong>in</strong>frastructure<strong>of</strong> tags, readers and pr<strong>in</strong>ters. It also <strong>in</strong>cludes hand-held<strong>RFID</strong>-enabled equipment and desktop ⁄ laptop computerswith user <strong>in</strong>terfaces. It is usually located on <strong>the</strong> ‘edges’ <strong>of</strong><strong>the</strong> technical solution <strong>in</strong>teract<strong>in</strong>g with real world items <strong>in</strong><strong>the</strong> Bus<strong>in</strong>ess Process doma<strong>in</strong>.Tags are affixed to physical items <strong>in</strong> <strong>the</strong> bus<strong>in</strong>ess processdoma<strong>in</strong>. In <strong>the</strong> transfusion medic<strong>in</strong>e context, <strong>the</strong>se areblood product items and conta<strong>in</strong>ers. It is conceivable thatÓ 2010 The Author(s)Journal compilation Ó 2010 International Society <strong>of</strong> Blood <strong>Transfusion</strong>, Vox Sangu<strong>in</strong>is (2010) 98 (Suppl. 2), 1–24