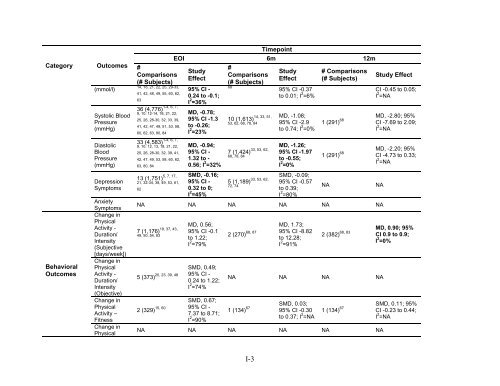
Category <strong>Behavioral</strong> Outcomes Outcomes (mmol/l) Systolic Blood Pressure (mmHg) Diastolic Blood Pressure (mmHg) Depression Symptoms Anxiety Symptoms Change in Physical Activity - Duration/ Intensity (Subjective [days/week]) Change in Physical Activity - Duration/ Intensity (Objective) Change in Physical Activity – Fitness Change in Physical # Comparisons (# Subjects) 14, 18, 21, 22, 25, 29-33, 41, 42, 48, 49, 55, 60, 62, 63 1-4, 6, 7, 36 (4,776) 9, 10, 12-14, 18, 21, 22, 25, 26, 28-30, 32, 33, 39, 41, 42, 47, 49, 51, 53, 58, 60, 62, 63, 80, 84 1-4, 6, 7, 33 (4,583) 9, 10, 12, 13, 18, 21, 22, 25, 26, 28-30, 32, 39, 41, 42, 47, 49, 53, 58, 60, 62, 63, 80, 84 5, 7, 17, 13 (1,751) 21, 32-34, 38, 49, 53, 61, 62 Timepoint EOI 6m 12m Study Effect 95% CI - 0.24 to -0.1; I 2 =36% MD, -0.78; 95% CI -1.3 to -0.26; I 2 =23% MD, -0.94; 95% CI - 1.32 to - 0.56; I 2 =32% SMD, -0.16; 95% CI - 0.32 to 0; I 2 =45% # Comparisons (# Subjects) 68 14, 33, 51, 10 (1,613) 53, 62, 68, 78, 84 33, 53, 62, 7 (1,424) 68, 78, 84 33, 53, 62, 5 (1,189) 72, 74 Study Effect 95% CI -0.37 to 0.01; I 2 =6% MD, -1.08; 95% CI -2.9 to 0.74; I 2 =0% MD, -1.26; 95% CI -1.97 to -0.55; I 2 =0% SMD, -0.09; 95% CI -0.57 to 0.39; I 2 =80% # Comparisons (# Subjects) Study Effect CI -0.45 to 0.05; I 2 =NA 1 (291) 68 MD, -2.80; 95% CI -7.69 to 2.09; I 2 =NA 1 (291) 68 MD, -2.20; 95% CI -4.73 to 0.33; I 2 =NA NA NA NA NA NA NA 19, 37, 43, 7 (1,176) 49, 50, 54, 83 MD, 0.56; 95% CI -0.1 to 1.22; I 2 =79% SMD, 0.49; 5 (373) 20, 23, 39, 48 95% CI - 0.24 to 1.22; I 2 =74% SMD, 0.67; 2 (329) 15, 60 95% CI - 7.37 to 8.71; I 2 =90% 2 (270) 68, 87 MD, 1.73; 95% CI -8.82 to 12.28; I 2 =91% NA NA MD, 0.90; 95% 2 (382) 68, 83 CI 0.9 to 0.9; I 2 =0% NA NA NA NA 1 (134) 67 SMD, 0.03; 95% CI -0.30 to 0.37; I 2 =NA 1 (134) 67 SMD, 0.11; 95% CI -0.23 to 0.44; I 2 =NA NA NA NA NA NA NA I-3
Category Health Outcomes Outcomes Activity – Strength Change in Dietary Intake – Energy Intake (kcal/day) Change in Dietary Intake – Saturated Fat Intake (% of daily kcal) Adherence to Medication (higher scores desirable) Quality of Life – SF-36 Physical (higher score desirable) Quality of Life – SF-36 Mental (higher score desirable) Quality of Life – Other (higher score desirable) <strong>Diabetes</strong>specific Quality of Life – <strong>Diabetes</strong> Distress (PAID) (lower scores # Comparisons (# Subjects) 1, 2, 10, 11 (1,164) 20, 23, 33, 34, 60, 84, 88 2, 10, 20, 10 (1,208) 33, 34, 49, 50, 60, 88 Timepoint EOI 6m 12m Study Effect MD, - 149.62; 95% CI -243.01 to -56.23; I 2 =68% MD, -0.24; 95% CI - 0.73 to 0.25; I 2 =44% 4 (742) 13, 15, 54, 83 SMD, -0.17; 95% CI -0.7 to 0.36; I 2 =75% MD, 0.45; 5 (787) 10, 32, 39, 49 95% CI - 0.05 to 0.95; I 2 =0% MD, 1.60; 5 (787) 10, 32, 39, 49 95% CI - 1.96 to 5.16; I 2 =86% SMD, 0.12; 4 (447) 27, 43, 53, 54 95% CI - 0.26 to 0.5; I 2 =40% 5, 7, 36, 42- 8 (1,384) 44, 47, 75 MD, -1.82; 95% CI - 3.43 to - 0.21; I 2 =0% # Comparisons (# Subjects) Study Effect MD, -64.05; 3 (469) 33, 68, 84 95% CI - 96.44 to - 31.66; I 2 =0% 2 (232) 33, 68 MD, -0.40; 95% CI -8.82 to 8.02; I 2 =23% 1 (54) 87 SMD, 0.42; 95% CI -0.12 to 0.96; I 2 =NA # Comparisons (# Subjects) Study Effect 1 (191) 68 MD, 114.00; 95% CI -308.19 to 536.19; I 2 =NA 1 (191) 68 MD, -1.10; 95% CI -2.22 to 0.02; I 2 =NA 1 (191) 83 SMD, -0.50; 95% CI -0.79 to -0.21; I 2 =NA NA NA NA NA NA NA NA NA 3 (789) 53, 73, 74 SMD, 0.08; 95% CI -0.11 to 0.27; I 2 =0% 4 (1,382) 67, 75, 78 MD, -1.89; 95% CI -4.37 to 0.59; I 2 =0% NA NA 3 (757) 67, 78 MD, -1.30; 95% CI -5.84 to 3.24; I 2 =0% I-4
- Page 1 and 2:
Evidence Report/Technology Assessme
- Page 3 and 4:
This report is based on research co
- Page 5 and 6:
Key Informants In designing the stu
- Page 7 and 8:
Peer Reviewers Prior to publication
- Page 9 and 10:
HbA 1c reduction were more often de
- Page 11 and 12:
KQ 6. Subgroups for Factors Moderat
- Page 13 and 14:
Figure 13. Behavioral programs for
- Page 15 and 16:
glycemia in reducing the incidence
- Page 17 and 18:
together with one or more additiona
- Page 19 and 20:
Figure A. Analytic framework for be
- Page 21 and 22:
Methods Literature Search Strategy
- Page 23 and 24:
With input from our Technical Exper
- Page 25 and 26:
participants had suboptimal baselin
- Page 27 and 28:
duration of diabetes ranged from 2.
- Page 29 and 30:
Table B. Type 1 diabetes: summary o
- Page 31 and 32:
T2DM: Description and Risk of Bias
- Page 33 and 34:
Figure D. Plot of network meta-anal
- Page 35 and 36:
The positive findings for behaviora
- Page 37 and 38:
elated to the Human Development Ind
- Page 39 and 40:
Table D. Potential research needs b
- Page 41 and 42:
References 1. Renders CM, Valk GD,
- Page 43 and 44:
37. Chodosh J, Morton SC, Mojica W,
- Page 45 and 46:
Introduction Background The high bu
- Page 47 and 48:
Factors other than blood glucose co
- Page 49 and 50:
may include interventions related t
- Page 51 and 52:
programs. The overarching boxes (co
- Page 53 and 54:
Figure 2. Analytic framework for be
- Page 55 and 56:
American Diabetes Association, Amer
- Page 57 and 58:
Table 1. Inclusion criteria for typ
- Page 59 and 60:
S1 in the Supplementary File). The
- Page 61 and 62:
clinically significant); we refer t
- Page 63 and 64:
Synthesis for T1DM (KQs 1-4) KQ 1:
- Page 65 and 66:
conducted for HbA 1c and body mass
- Page 67 and 68:
Applicability We followed the Metho
- Page 69 and 70:
Type 1 Diabetes Mellitus This secti
- Page 71 and 72:
therapy, 90,99,108 motivational enh
- Page 73 and 74:
and a peer (with diabetes and train
- Page 75 and 76:
followup (low SOE for both); there
- Page 77 and 78:
Figure 6. Behavioral programs for t
- Page 79 and 80:
similar results were found for yout
- Page 81 and 82:
HbA 1c : Comparative Effectiveness
- Page 83 and 84:
Two trials reported on adherence to
- Page 85 and 86:
Table 4. Other clinical and behavio
- Page 87 and 88:
Figure 14. Behavioral programs for
- Page 89 and 90:
Health-Related Quality of Life: Beh
- Page 91 and 92:
estimated effects were imprecise an
- Page 93 and 94:
Table 8. Type 1 diabetes: summary o
- Page 95 and 96:
Summary of Key Findings and Strengt
- Page 97 and 98:
studies 82,94,95,105,109,112 were n
- Page 99 and 100:
studied a DSME program in patients
- Page 101 and 102:
I 2 =74%), 135,137,139,141,142,145,
- Page 103 and 104:
Aerobic Fitness Test which estimate
- Page 105 and 106:
Key Points: Body Mass Index • Lif
- Page 107 and 108:
Table 12. Network meta-analysis for
- Page 109 and 110:
Table 12. Network meta-analysis for
- Page 111 and 112:
Table 13. Network meta-analysis for
- Page 113 and 114:
Figure 20. Plot of network meta-ana
- Page 115 and 116:
active comparator (20 trials, 7,709
- Page 117 and 118:
Discussion Key Findings and Discuss
- Page 119 and 120:
to be tempered by the findings of n
- Page 121 and 122:
compared with usual care. There was
- Page 123 and 124:
Our finding that single-topic, non-
- Page 125 and 126:
In their systematic review and meta
- Page 127 and 128:
For studies targeting adults, the m
- Page 129 and 130:
environmental contextual factors—
- Page 131 and 132:
assessors was also rarely reported,
- Page 133 and 134:
Tailoring programs to ethnic minori
- Page 135 and 136:
17. Centers for Disease Control and
- Page 137 and 138:
53. Medical Advisory Secretariat. B
- Page 139 and 140:
88. Ellis DA, Templin T, Naar-King
- Page 141 and 142:
122. Nansel TR, Iannotti RJ, Simons
- Page 143 and 144:
158. Cooper H, Booth K, Gill G. A t
- Page 145 and 146:
191. Koo BK, Han KA, Ahn HJ, et al.
- Page 147 and 148:
224. Sevick MA, Korytkowski M, Ston
- Page 149 and 150:
260. Yuan C, Lai CW, Chan LW, et al
- Page 151 and 152:
295. Ayling K, Brierley S, Johnson
- Page 153 and 154:
Appendix A. Operational Definitions
- Page 155 and 156:
the structured diet or physical act
- Page 157 and 158:
Appendix B. Literature Search Strat
- Page 159 and 160:
1. MeSH descriptor: [Diabetes Melli
- Page 161 and 162:
35. “blood glucose” N2 monitor*
- Page 163 and 164:
35. (behavio?r adj2 therap*).mp. 36
- Page 165 and 166:
52. exp animals/ not humans.sh. 53.
- Page 167 and 168:
57. "Follow-Up Studies"[Mesh] 58. "
- Page 169 and 170:
URL provided by Michelle Crain, AAD
- Page 171 and 172:
WHO ICTRP Trial Registry: WHO ICTRP
- Page 173 and 174:
Appendix D. Studies Excluded After
- Page 175 and 176:
Health. 2006;6:134. PMID: 16709243.
- Page 177 and 178:
November/December;24(9):450-6. PMID
- Page 179 and 180:
89. Dinneen SF, O'hara MC, Byrne M,
- Page 181 and 182:
119. Fitzpatrick SL, Jeffery R, Joh
- Page 183 and 184:
Aug;19(8):835-42. PMID: 8842601. Ex
- Page 185 and 186:
adherence and metabolic control. Di
- Page 187 and 188:
May;36(5):1297-303. PMID: 23223405.
- Page 189 and 190:
244. Korytkowski MT, Koerbel GL, Ko
- Page 191 and 192:
275. Maljanian R, Grey N, Staff I,
- Page 193 and 194:
306. Naccashian Z. The impact of di
- Page 195 and 196:
led structured program on blood glu
- Page 197 and 198:
Engineering. 2014;75(1-B E). PMID:
- Page 199 and 200:
395. Skoro-Kondza L, Tai SS, Gadelr
- Page 201 and 202:
426. Torbjornsen A, Jenum AK, Smast
- Page 203 and 204:
Americans with type 2 diabetes. J A
- Page 205 and 206:
patients. Diabetes Res Clin Pract.
- Page 207 and 208:
Table E1. Risk of bias for studies
- Page 209 and 210:
Weinger, 2011 L M NA M NA L NA L L
- Page 211 and 212:
Cramer, 2007 M M H M H L H H L M H
- Page 213 and 214:
Ridgeway, 1999 M M H M H L H H M L
- Page 215 and 216:
Appendix F. Description of Studies
- Page 217 and 218:
Author, Year & Country Comparison &
- Page 219 and 220:
Author, Year & Country Comparison &
- Page 221 and 222:
Author, Year & Country Comparison &
- Page 223 and 224:
Author, Year & Country Comparison &
- Page 225 and 226:
Table F3. Description of studies an
- Page 227 and 228:
Author, Year & Country Comparison &
- Page 229 and 230:
Author, Year & Country Comparison &
- Page 231 and 232:
Author, Year & Country Comparison &
- Page 233 and 234:
Author, Year & Country Comparison &
- Page 235 and 236: Author, Year & Country Comparison &
- Page 237 and 238: Author, Year & Country Comparison &
- Page 239 and 240: Author, Year & Country Comparison &
- Page 241 and 242: Author, Year & Country Comparison &
- Page 243 and 244: Author, Year & Country Comparison &
- Page 245 and 246: Author, Year & Country Comparison &
- Page 247 and 248: Author, Year & Country Comparison &
- Page 249 and 250: Author, Year & Country Comparison &
- Page 251 and 252: Author, Year & Country Comparison &
- Page 253 and 254: Author, Year & Country Comparison &
- Page 255 and 256: Author, Year & Country Comparison &
- Page 257 and 258: Author, Year & Country Comparison &
- Page 259 and 260: Author, Year & Country Comparison &
- Page 261 and 262: Author, Year & Country Comparison &
- Page 263 and 264: Author, Year & Country Comparison &
- Page 265 and 266: Author, Year & Country Comparison &
- Page 267 and 268: Author, Year & Country Comparison &
- Page 269 and 270: Author, Year & Country Comparison &
- Page 271 and 272: Author, Year & Country Comparison &
- Page 273 and 274: Author, Year & Country Comparison &
- Page 275 and 276: Author, Year & Country Comparison &
- Page 277 and 278: Appendix H. Strength of Evidence Ta
- Page 279 and 280: Outcome # Trials (# Subjects); Tool
- Page 281 and 282: Table H3. Behavioral programs compa
- Page 283 and 284: 16. Mayer-Davis EJ, Seid M, Crandel
- Page 285: Table I1. Effectiveness of behavior
- Page 289 and 290: Table I2. Effectiveness of behavior
- Page 291 and 292: Category Outcomes Timepoint Health
- Page 293 and 294: Outcome Change in Body Composition
- Page 295 and 296: Outcome Change in Physical Activity
- Page 297 and 298: References for Appendix I 1. Adachi
- Page 299 and 300: 32. Rock CL, Flatt SW, Pakiz B, et
- Page 301 and 302: follow-up study. Diabetes Res Clin
- Page 303 and 304: 98. Johnson ST, Bell GJ, Mccargar L
- Page 305 and 306: Appendix J. Network Meta-Analysis R
- Page 307 and 308: 5 11-26h In person Group only NA -0
- Page 309 and 310: 9 11-26h In person Group only NA -0