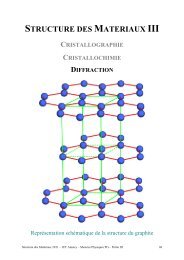
Structure des Matériaux Partie I, Cristallographie - IUT Annecy
Structure des Matériaux Partie I, Cristallographie - IUT Annecy
Structure des Matériaux Partie I, Cristallographie - IUT Annecy
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Cuivre, Cu<br />
cubique, Fm3 m<br />
a = 3.61 Å<br />
Cu 0 0 0<br />
(≡ quatre atomes de Cu en 0 0 0, 0 ½ ½, ½ 0 ½, ½ ½ 0)<br />
V = 47.05 Å 3<br />
Z = 4<br />
M r = 63.55 g mol -1<br />
D x = 8.97 Mg m -3 (Dm = 8.94 Mg m -3 )<br />
Cu 3 Au<br />
cubique, Pm3 m<br />
a = 3.74 Å<br />
V = 52.31 Å 3<br />
Au 0 0 0 (bleu)<br />
Cu 0 ½ ½ (rouge)<br />
(≡ trois atomes de Cu en 0 ½ ½, ½ 0 ½, ½ ½ 0)<br />
Z = 1<br />
M r = 387.6 g mol -1<br />
D x = 12.30 Mg m -3<br />
<strong>Structure</strong> <strong>des</strong> <strong>Matériaux</strong> 2011 – <strong>IUT</strong> <strong>Annecy</strong> – Mesures Physiques PG – <strong>Partie</strong> I 33