Procedure for assessing the acceptability, in principle, of vaccines ...
Procedure for assessing the acceptability, in principle, of vaccines ...
Procedure for assessing the acceptability, in principle, of vaccines ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
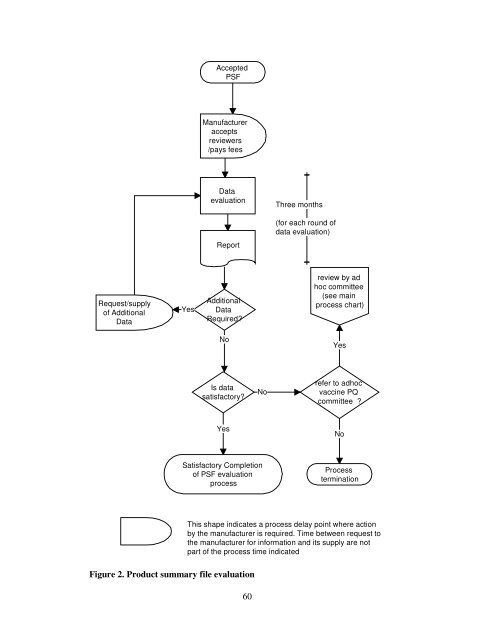
Request/supply<br />
<strong>of</strong> Additional<br />
Data<br />
Yes<br />
Accepted<br />
PSF<br />
Manufacturer<br />
accepts<br />
reviewers<br />
/pays fees<br />
Data<br />
evaluation<br />
Report<br />
Additional<br />
Data<br />
Required?<br />
No<br />
Is data<br />
satisfactory?<br />
Yes<br />
Satisfactory Completion<br />
<strong>of</strong> PSF evaluation<br />
process<br />
Figure 2. Product summary file evaluation<br />
60<br />
No<br />
Three months<br />
(<strong>for</strong> each round <strong>of</strong><br />
data evaluation)<br />
review by ad<br />
hoc committee<br />
(see ma<strong>in</strong><br />
process chart)<br />
Yes<br />
refer to adhoc<br />
vacc<strong>in</strong>e PQ<br />
committee ?<br />
No<br />
Process<br />
term<strong>in</strong>ation<br />
This shape <strong>in</strong>dicates a process delay po<strong>in</strong>t where action<br />
by <strong>the</strong> manufacturer is required. Time between request to<br />
<strong>the</strong> manufacturer <strong>for</strong> <strong>in</strong><strong>for</strong>mation and its supply are not<br />
part <strong>of</strong> <strong>the</strong> process time <strong>in</strong>dicated