Procedure for assessing the acceptability, in principle, of vaccines ...
Procedure for assessing the acceptability, in principle, of vaccines ...
Procedure for assessing the acceptability, in principle, of vaccines ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
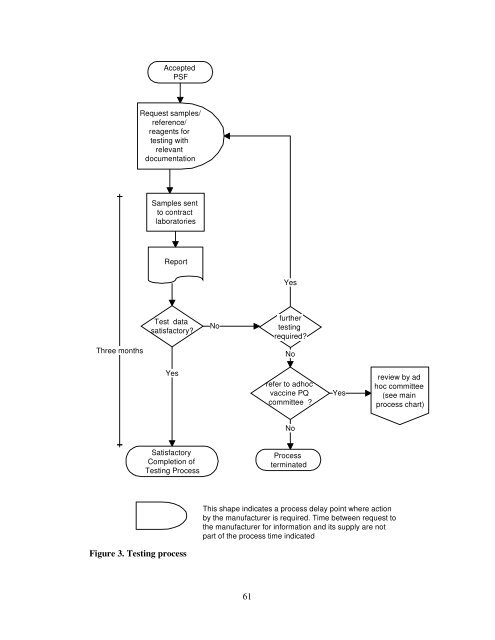
Three months<br />
Accepted<br />
PSF<br />
Request samples/<br />
reference/<br />
reagents <strong>for</strong><br />
test<strong>in</strong>g with<br />
relevant<br />
documentation<br />
Samples sent<br />
to contract<br />
laboratories<br />
Report<br />
Test data<br />
satisfactory?<br />
Yes<br />
Satisfactory<br />
Completion <strong>of</strong><br />
Test<strong>in</strong>g Process<br />
Figure 3. Test<strong>in</strong>g process<br />
No<br />
61<br />
Yes<br />
fur<strong>the</strong>r<br />
test<strong>in</strong>g<br />
required?<br />
No<br />
refer to adhoc<br />
vacc<strong>in</strong>e PQ<br />
committee ?<br />
No<br />
Process<br />
term<strong>in</strong>ated<br />
Yes<br />
This shape <strong>in</strong>dicates a process delay po<strong>in</strong>t where action<br />
by <strong>the</strong> manufacturer is required. Time between request to<br />
<strong>the</strong> manufacturer <strong>for</strong> <strong>in</strong><strong>for</strong>mation and its supply are not<br />
part <strong>of</strong> <strong>the</strong> process time <strong>in</strong>dicated<br />
review by ad<br />
hoc committee<br />
(see ma<strong>in</strong><br />
process chart)