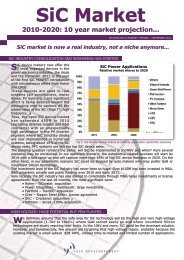
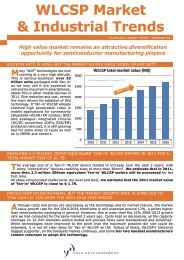
OCTOBER 2010 issue n°100 THE DISRUPTIVE SEMICONDUCTOR TECHNOLOGIES MAGAZINE Copyrights © Yole Développement SA. All rights reserved - Recycled paper 10
OCTOBER 2010 issue n°100 THE DISRUPTIVE SEMICONDUCTOR TECHNOLOGIES MAGAZINE MICROFLUIDICS New microfluidic device helps identify and capture breast cancer cells Researchers have developed a new disposable device that could help identify patients with advanced breast cancer awaiting drug <strong>the</strong>rapy with trastuzumab, and capture cancer cells that have an abnormal amount of <strong>the</strong> protein HER2. Benjamin Thierry, of <strong>the</strong> Ian Wark Research Institute at <strong>the</strong> University of South Australia, along with his colleagues, has created an elastic, disposable microfluidic device that can efficiently catch <strong>the</strong> protein HER2 – which is <strong>the</strong> tyrosine kinase human epidermal growth factor receptor 2 – found in "aggressive breast cancers with poor prognosis." The only way to find out HER2 status in breast cancer patients now is through fluorescence in situ hybridization (FISH) or immunohistochemistry, both of which require biopsies. The problem with biopsybased testing is that it could lead to treatment that is ineffective, because <strong>the</strong> HER2 status of <strong>the</strong> primary tumor in 20 percent of breast cancers differs from <strong>the</strong> HER2 status in that of a metastatic tumor. This information has led to <strong>the</strong> idea of isolating circulating tumor cells (CTCs), but this is difficult because circulating tumor cells exist at very low ratios of 1 to 10 per billion blood cells. Establishing HER2 status is very critical because HER2 positive breast cancer patients have benefitted from drug <strong>the</strong>rapy with trastuzumab (Herceptin), which are humanized monoclonal antibodies against HER2. It also decreases recurrence risk by half. Also, <strong>the</strong> side effects and cost makes it a priority to identify patients who are HER2 positive. Now, Thierry and his colleagues' device has <strong>the</strong> ability to detect <strong>the</strong> status of HER2 and help capture circulating tumor cells. The device is made of organic silicon found in most contact lenses, and polydimethylsiloxane (PDMS), which are shampoos that are transparent, permeable to gasses, and compatible with soft molding techniques. The device also contains a polymeric layer on its surface, which has a large number of reactive molecules in it. Through <strong>the</strong> use of a plasma-based polymerization process, this layer can attach to proteins that are able to catch cancer cells while leaving normal blood cells alone. Thierry's device showed an 80 percent immunocapture efficacy of HER2 positive cells, and is also cheaper and easier to make than previous microfabricated devices, which were developed to bind to cells of epi<strong>the</strong>lial tissues where <strong>the</strong> cancer originated, ultimately isolating CTCs. www.unisa.edu.au Lockheed Martin and ZyGEM accelerate DNA identification for law enforcement, homeland security and defense communities New, fast sample-to-answer DNA analysis platform previewed at 2010 Biometric Consortium conference. Lockheed Martin and ZyGEM Corp. Ltd. are unveiling a new DNA analysis system that has potential to transform how law enforcement, homeland security and defense communities use DNA-based identification in support of <strong>the</strong>ir missions. The prototype, called RapI.D., will be previewed at a 2010 Biometric Consortium Conference special session on Rapid DNA. Developed in collaboration with ZyGEM as a Lockheed Martin Technology Innovation Initiative, RapI.D. leverages <strong>the</strong> latest in microfluidic research and development to accelerate <strong>the</strong> DNA identification process – essentially building a laboratory on a small, single chip that reduces <strong>the</strong> processing steps and time needed for analysis. The RapI.D. platform is currently in prototype at ZyGEM's Charlottesville, Va., MicroLab laboratories, with a Beta version expected to be released for testing in select laboratories early next year. The cost and complexity of current forensic DNA analysis methods has contributed to significant processing backlogs throughout <strong>the</strong> criminal justice system. Data from <strong>the</strong> Justice Department's FY 2009 Forensic DNA Backlog Reduction Program suggest that <strong>the</strong> backlog has increased in recent years, indicating that <strong>the</strong> 2008 backlogged cases reported by state and local government applicants for funding under <strong>the</strong> program had nearly tripled compared to comparable data from 2005. www.lockheedmartin.com www.zygem.com Pacific Biosciences to raise up to $230 M in IPO Pacific Biosciences of California Inc said it expects to raise up to $230 million in an initial public offering of its common stock. The Menlo Park, California-based company expects to offer 12.5 million shares at $15-$17 each, with an overallotment option of 1.9 million shares, it said in an amended filing. The life sciences company, which designs machines to speed up DNA sequencing in labs, sees net proceeds of $182.5 million, assuming <strong>the</strong> offering priced at $16 per share, <strong>the</strong> mid-point of <strong>the</strong> range. The company plans to invest about $60-$70 million of <strong>the</strong> proceeds in its technology platform called single molecule, realtime, or SMRT technology. Pacific Biosciences initially filed for an IPO in August. The amended filing detailed <strong>the</strong> expected pricing of <strong>the</strong> common stock sale. JP Morgan, Morgan Stanley, Deutsche Bank and Piper Jaffray are underwriting <strong>the</strong> IPO, <strong>the</strong> company said. Pacific Biosciences plans to list its shares under <strong>the</strong> ticker symbol "PACB" on Nasdaq. www.pacificbiosciences.com FluimediX closes financing round Funding Supports continued development of <strong>the</strong> NanoCycler platform to include a Warfarin Diagnostic Test. FluimediX announced that it has received funding for <strong>the</strong> continued development of <strong>the</strong> NanoCycler, a diagnostic platform for <strong>the</strong> measurement of relevant genetic mutations in patients. The proceeds of this financing round will be dedicated to developing a diagnostic test for Warfarin metabolism. The investment round was led by SEED Capital Denmark, a leading early stage investor managing more than € 200 million in total assets. Existing shareholders also participated in <strong>the</strong> financing. The size of <strong>the</strong> round was not disclosed. Tomas Ussing, CEO and founder of FluimediX, commented: “Firstly, it is comprised of low cost, wellestablished components. Secondly, <strong>the</strong> use of plastic disposables enables extremely competitive molecular diagnostic testing, and will thus bring this type of testing closer to <strong>the</strong> patient. Thirdly, <strong>the</strong> built-in broad versatility of <strong>the</strong> NanoCycler device will allow o<strong>the</strong>r tests to be transferred to or developed for <strong>the</strong> NanoCycler platform in <strong>the</strong> future.” The use of a diagnostic test for Warfarin metabolism would deliver important clinical benefits and cost savings to <strong>the</strong> healthcare system. Approximately two million people start taking Warfarin each year in <strong>the</strong> U.S. Unfortunately, <strong>the</strong> optimal dose differs significantly due to patient-to-patient genetic variation in how quickly Warfarin is metabolized, and <strong>the</strong> medical consequences of under- or over-dosing <strong>the</strong> drug can be severe. Roughly one-third of <strong>the</strong> population carries a genetic mutation that results in slow metabolism of <strong>the</strong> drug, so <strong>the</strong> identification of those patients who have this mutation could help avoid <strong>the</strong> consequences of under- or over-dosing. FX device lunch-box www.fluimedix.com FX chip Copyrights © Yole Développement SA. All rights reserved - Recycled paper 11