CDC Article-US Medical Eligibility Criteria for Contraceptive Use, 2010
CDC Article-US Medical Eligibility Criteria for Contraceptive Use, 2010
CDC Article-US Medical Eligibility Criteria for Contraceptive Use, 2010
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
52 MMWR June 18, <strong>2010</strong><br />
Appendix E<br />
Classifications <strong>for</strong> Intrauterine Devices<br />
Classifications <strong>for</strong> intrauterine devices (IUDs) are <strong>for</strong> the<br />
levonorgestrel-releasing (20 μg/24 hours) IUD and the copperbearing<br />
IUD (Box). IUDs do not protect against sexually<br />
transmitted infections (STIs) or human immunodeficiency<br />
virus (HIV).<br />
BOX. Categories <strong>for</strong> Classifying Intrauterine Devices<br />
1 = A condition <strong>for</strong> which there is no restriction <strong>for</strong> the use of the contraceptive method.<br />
2 = A condition <strong>for</strong> which the advantages of using the method generally outweigh the theoretical or proven risks.<br />
3 = A condition <strong>for</strong> which the theoretical or proven risks usually outweigh the advantages of using the method.<br />
4 = A condition that represents an unacceptable health risk if the contraceptive method is used.<br />
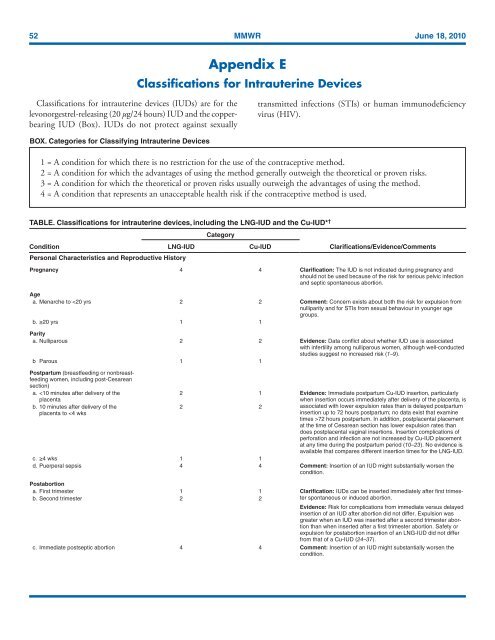
TABLE. Classifications <strong>for</strong> intrauterine devices, including the LNG-IUD and the Cu-IUD* †<br />
Condition<br />
LNG-IUD<br />
Personal Characteristics and Reproductive History<br />
Category<br />
Cu-IUD<br />
Clarifications/Evidence/Comments<br />
Pregnancy 4 4 Clarification: The IUD is not indicated during pregnancy and<br />
should not be used because of the risk <strong>for</strong> serious pelvic infection<br />
and septic spontaneous abortion.<br />
Age<br />
a. Menarche to