Nucleic Acid Analysis with UV-vis and NMR - Spectroscopy
Nucleic Acid Analysis with UV-vis and NMR - Spectroscopy
Nucleic Acid Analysis with UV-vis and NMR - Spectroscopy
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
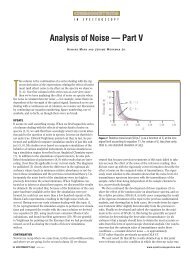
26 <strong>Spectroscopy</strong> 24(11) November 2009 www.spectroscopyonline.com<br />
• FDA has now set postinspection<br />
deadlines for responses by organizations.<br />
The response time for firms to<br />
respond has been halved to 15 working<br />
days. This is not just to send a letter<br />
to begin a cozy correspondence <strong>with</strong><br />
the agency but to have a complete<br />
response to all the 483 observations.<br />
If the compliance problems are significant,<br />
the timeframe for the response is<br />
critical <strong>and</strong> the response must be full,<br />
adequate, <strong>and</strong> complete. The letter will<br />
not be able to show that the problems<br />
have been resolved for all but the simplest<br />
observations but will demonstrate<br />
how they will be resolved <strong>and</strong> in what<br />
timeframe. If a response is not received<br />
in this time or the response is inadequate,<br />
then the agency can start work<br />
on a warning letter or other enforcement<br />
action. We’ll take a look at ways<br />
to respond to a 483 or warning letter in<br />
a later section of this column.<br />
An important point to underst<strong>and</strong><br />
here is that the FDA now takes the<br />
systems-based approach to inspections<br />
<strong>and</strong> coupled <strong>with</strong> ICH Q10 on<br />
pharmaceutical quality systems (6)<br />
you will need a different approach to<br />
respond to 483 observations. If there<br />
is an observation that has an impact<br />
outside of the area inspected, you will<br />
be expected to fix it as part of the systems-based<br />
approach. Remember, the<br />
wording at the end of virtually every<br />
warning letter on the FDA website:<br />
“this letter is not intended to be an<br />
all-inclusive list of the violations at<br />
your facility. It is your responsibility<br />
to ensure compliance <strong>with</strong> applicable<br />
laws <strong>and</strong> regulations administered by<br />
FDA.” However, there is an onus on<br />
the inspector to discuss findings <strong>with</strong><br />
management to minimize surprises<br />
<strong>and</strong> errors when issuing the 483 at the<br />
inspection closing meeting.<br />
• The warning letter issuing process<br />
is being streamlined <strong>and</strong> speeded<br />
up by the FDA. Instead of the FDA<br />
lawyers reviewing all warning letters,<br />
only those where there are significant<br />
legal issues will now get the legal eagle<br />
eye. The agency will not delay issuing<br />
a warning letter if you are late in<br />
responding to the 483. Even if your<br />
response to the 483 is perfect, the FDA<br />
does “not plan to routinely include a<br />
response on the apparent adequacy<br />
of the firm’s corrective actions in the<br />
warning letter (4).” Instead, the agency<br />
will respond to evaluate both your<br />
response to the 483 <strong>and</strong> the warning<br />
letter together. Therefore, the message<br />
is clear to all: Your 483 response must<br />
be timely <strong>and</strong> complete, or else you<br />
could end up <strong>with</strong> a warning letter on<br />
the front doormat pretty quickly afterwards.<br />
• Next, the FDA will prioritize<br />
enforcement follow-up. After a warning<br />
letter is issued or a major product<br />
recall occurs, the FDA will make it a<br />
priority to follow up promptly <strong>with</strong><br />
appropriate action, such as an inspection<br />
or investigation to assess whether<br />
or not a company has made required<br />
changes in its compliance practices.<br />
• In the past, it was not uncommon<br />
for a company to have a number<br />
of warning letters after several inspections;<br />
it may be the companies