4. Clinical Guidelines for Liver Transplantation (PDF) - British ...
4. Clinical Guidelines for Liver Transplantation (PDF) - British ...
4. Clinical Guidelines for Liver Transplantation (PDF) - British ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
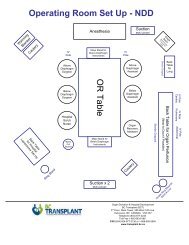
Guidebook <strong>for</strong> the Solid Organ Transplant Programme Chapter 4<br />
Appendix F<br />
BCT FAX FORM: APPLICATION FOR SIROLIMUS LIVER TRANSPLANT<br />
RECIPIENTS<br />
Please complete & fax to Dawn Strong <strong>for</strong> Dr. YOSHIDA at B.C. Transplant (604)- 877-2111<br />
DATE:<br />
TO:<br />
FROM:<br />
Dr. Eric Yoshida<br />
Medical Director, <strong>Liver</strong> Transplant Program, BCT<br />
BCT #:_______________________<br />
Name: Last: _________________________________ First: ______________________<br />
Hospital: _________________________ Hepatologist: _________________________<br />
Indications <strong>for</strong> Sirolimus Use:<br />
1. Patient has developed calcineurin toxicity despite blood concentrations within therapeutic range.<br />
Cyclosporine concentration _________ng/mL (date________)<br />
Tacrolimus concentration __________ng/mL (date________)<br />
a) biopsy-proven severe nephrotoxicity. Increase in serum creatinine must be 50% above baseline.<br />
Biopsy result ____________________________________(date_________)<br />
Baseline serum creatinine ____________________µmol/L (date_________)<br />
Current serum creatinine ____________________µmol/L (date_________)<br />
b) Neurotoxicity (describe reaction) ______________________________________ (date______)<br />
2. Patient has developed calcineurin inhibitor intolerance: hypersensitivity reaction or microangiopathy<br />
Cyclosporine (date_________) Tacrolimus (date______)<br />
3. Recurrent (≥ 2) biopsy-proven, acute rejection, while on calcineurin inhibitors despite blood<br />
concentrations within the therapeutic range.<br />
Biopsy result: ___________________________ (date_______)<br />
Biopsy result: ___________________________ (date________)<br />
Cyclosporine concentration: _________________mol/L (date_________)<br />
Tacrolimus concentration: _________________mol/L (date_________)<br />
<strong>4.</strong> Maintenance immunosuppression following steroid-resistant rejection. ATG use __________(date)<br />
5. Renal dysfunction due to: hepato-renal syndrome or post transplant acute tubular necrosis<br />
6. Acute rejection not requiring ATG, but sirolimus is necessary <strong>for</strong> maintenance<br />
immunosuppression.<br />
Physician’s Signature: __________________________ Date: ____________<br />
Approval by BCT: _____________________________ Date: ____________<br />
FEB/2010<br />
Chapter 4 – <strong>Clinical</strong> <strong>Guidelines</strong> <strong>for</strong> <strong>Liver</strong> <strong>Transplantation</strong> – July, 2010 Page 47<br />
See Page 1 <strong>for</strong> disclaimer