4. Clinical Guidelines for Liver Transplantation (PDF) - British ...
4. Clinical Guidelines for Liver Transplantation (PDF) - British ...
4. Clinical Guidelines for Liver Transplantation (PDF) - British ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
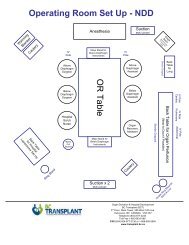
Guidebook <strong>for</strong> the Solid Organ Transplant Programme Chapter 4<br />
Appendix S (con’t)<br />
Recommended Follow-up Testing <strong>for</strong> Recipients Transplanted under Risk <strong>for</strong> Viral<br />
Mediated Disease Transmission<br />
NOTICE TO TRANSPLANT RECIPIENT MEDICAL TEAM:<br />
The following protocol has been approved by the BC Transplant Medical Advisory Committee<br />
(MAC) as a course of action <strong>for</strong> follow-up of recipients transplanted at risk <strong>for</strong> HIV and/or Hepatitis:<br />
IT IS RECOMMENDED THAT RECIPIENTS ARE RETESTED FOR HIV, HEPATITIS B AND<br />
HEPATITIS C at:<br />
• 4 weeks<br />
• 8 weeks<br />
• 6 months<br />
• 1 year<br />
Recommended Test Methods**:<br />
HIV - Conventional antibody testing<br />
HBV - HBsAg and HBcIgM<br />
HCV - Conventional antibody testing<br />
**NOTE: If there is clinical or epidemiological evidence to suggest a patient may have become<br />
infected with any of these viruses and antibody tests are negative, then PCR testing should be<br />
discussed with the medical team.<br />
Chapter 4 – <strong>Clinical</strong> <strong>Guidelines</strong> <strong>for</strong> <strong>Liver</strong> <strong>Transplantation</strong> – July, 2010 Page 66<br />
See Page 1 <strong>for</strong> disclaimer