Use of oxytocin and misoprostol for induction or ... - POPPHI
Use of oxytocin and misoprostol for induction or ... - POPPHI
Use of oxytocin and misoprostol for induction or ... - POPPHI
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
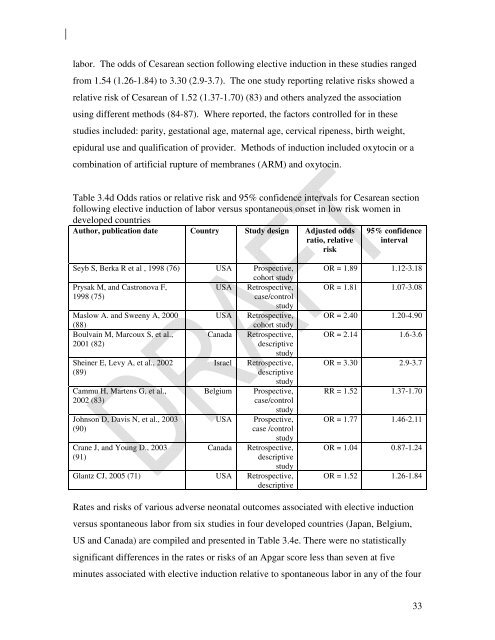
lab<strong>or</strong>. The odds <strong>of</strong> Cesarean section following elective <strong>induction</strong> in these studies ranged<br />
from 1.54 (1.26-1.84) to 3.30 (2.9-3.7). The one study rep<strong>or</strong>ting relative risks showed a<br />
relative risk <strong>of</strong> Cesarean <strong>of</strong> 1.52 (1.37-1.70) (83) <strong>and</strong> others analyzed the association<br />
using different methods (84-87). Where rep<strong>or</strong>ted, the fact<strong>or</strong>s controlled <strong>f<strong>or</strong></strong> in these<br />
studies included: parity, gestational age, maternal age, cervical ripeness, birth weight,<br />
epidural use <strong>and</strong> qualification <strong>of</strong> provider. Methods <strong>of</strong> <strong>induction</strong> included <strong>oxytocin</strong> <strong>or</strong> a<br />
combination <strong>of</strong> artificial rupture <strong>of</strong> membranes (ARM) <strong>and</strong> <strong>oxytocin</strong>.<br />
Table 3.4d Odds ratios <strong>or</strong> relative risk <strong>and</strong> 95% confidence intervals <strong>f<strong>or</strong></strong> Cesarean section<br />
following elective <strong>induction</strong> <strong>of</strong> lab<strong>or</strong> versus spontaneous onset in low risk women in<br />
developed countries<br />
Auth<strong>or</strong>, publication date Country Study design Adjusted odds<br />
ratio, relative<br />
risk<br />
95% confidence<br />
interval<br />
Seyb S, Berka R et al , 1998 (76) USA Prospective,<br />
coh<strong>or</strong>t study<br />
Prysak M, <strong>and</strong> Castronova F,<br />
USA Retrospective,<br />
1998 (75)<br />
case/control<br />
study<br />
Maslow A. <strong>and</strong> Sweeny A, 2000 USA Retrospective,<br />
(88)<br />
Boulvain M, Marcoux S, et al.,<br />
2001 (82)<br />
Sheiner E, Levy A, et al., 2002<br />
(89)<br />
Cammu H, Martens G, et al.,<br />
2002 (83)<br />
Johnson D, Davis N, et al., 2003<br />
(90)<br />
Crane J, <strong>and</strong> Young D., 2003<br />
(91)<br />
Canada<br />
Israel<br />
Belgium<br />
USA<br />
Canada<br />
coh<strong>or</strong>t study<br />
Retrospective,<br />
descriptive<br />
study<br />
Retrospective,<br />
descriptive<br />
study<br />
Prospective,<br />
case/control<br />
study<br />
Prospective,<br />
case /control<br />
study<br />
Retrospective,<br />
descriptive<br />
study<br />
Glantz CJ, 2005 (71) USA Retrospective,<br />
descriptive<br />
OR = 1.89 1.12-3.18<br />
OR = 1.81 1.07-3.08<br />
OR = 2.40 1.20-4.90<br />
OR = 2.14 1.6-3.6<br />
OR = 3.30 2.9-3.7<br />
RR = 1.52 1.37-1.70<br />
OR = 1.77 1.46-2.11<br />
OR = 1.04 0.87-1.24<br />
OR = 1.52 1.26-1.84<br />
Rates <strong>and</strong> risks <strong>of</strong> various adverse neonatal outcomes associated with elective <strong>induction</strong><br />
versus spontaneous lab<strong>or</strong> from six studies in four developed countries (Japan, Belgium,<br />
US <strong>and</strong> Canada) are compiled <strong>and</strong> presented in Table 3.4e. There were no statistically<br />
significant differences in the rates <strong>or</strong> risks <strong>of</strong> an Apgar sc<strong>or</strong>e less than seven at five<br />
minutes associated with elective <strong>induction</strong> relative to spontaneous lab<strong>or</strong> in any <strong>of</strong> the four<br />
33