Nitrazine Paper - Point of Care Testing - Duke University
Nitrazine Paper - Point of Care Testing - Duke University
Nitrazine Paper - Point of Care Testing - Duke University
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
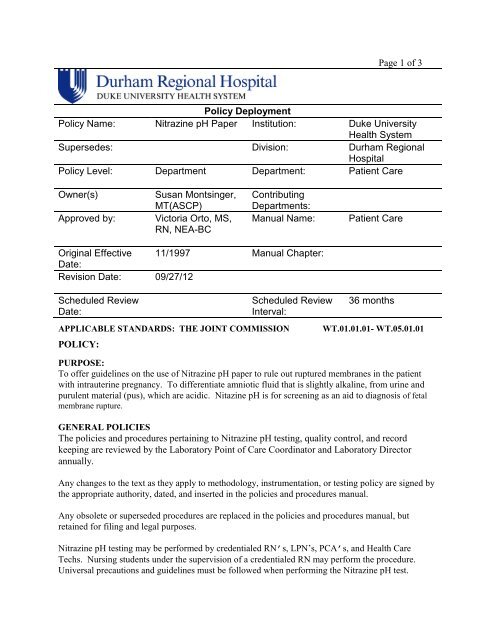
Page 1 <strong>of</strong> 3<br />
Policy Deployment<br />
Policy Name: <strong>Nitrazine</strong> pH <strong>Paper</strong> Institution: <strong>Duke</strong> <strong>University</strong><br />
Health System<br />
Supersedes: Division: Durham Regional<br />
Hospital<br />
Policy Level: Department Department: Patient <strong>Care</strong><br />
Owner(s)<br />
Approved by:<br />
Susan Montsinger,<br />
MT(ASCP)<br />
Victoria Orto, MS,<br />
RN, NEA-BC<br />
Contributing<br />
Departments:<br />
Manual Name:<br />
Patient <strong>Care</strong><br />
Original Effective 11/1997 Manual Chapter:<br />
Date:<br />
Revision Date: 09/27/12<br />
Scheduled Review<br />
Date:<br />
APPLICABLE STANDARDS: THE JOINT COMMISSION<br />
POLICY:<br />
Scheduled Review<br />
Interval:<br />
36 months<br />
WT.01.01.01- WT.05.01.01<br />
PURPOSE:<br />
To <strong>of</strong>fer guidelines on the use <strong>of</strong> <strong>Nitrazine</strong> pH paper to rule out ruptured membranes in the patient<br />
with intrauterine pregnancy. To differentiate amniotic fluid that is slightly alkaline, from urine and<br />
purulent material (pus), which are acidic. Nitazine pH is for screening as an aid to diagnosis <strong>of</strong> fetal<br />
membrane rupture.<br />
GENERAL POLICIES<br />
The policies and procedures pertaining to <strong>Nitrazine</strong> pH testing, quality control, and record<br />
keeping are reviewed by the Laboratory <strong>Point</strong> <strong>of</strong> <strong>Care</strong> Coordinator and Laboratory Director<br />
annually.<br />
Any changes to the text as they apply to methodology, instrumentation, or testing policy are signed by<br />
the appropriate authority, dated, and inserted in the policies and procedures manual.<br />
Any obsolete or superseded procedures are replaced in the policies and procedures manual, but<br />
retained for filing and legal purposes.<br />
<strong>Nitrazine</strong> pH testing may be performed by credentialed RN’s, LPN’s, PCA’s, and Health <strong>Care</strong><br />
Techs. Nursing students under the supervision <strong>of</strong> a credentialed RN may perform the procedure.<br />
Universal precautions and guidelines must be followed when performing the <strong>Nitrazine</strong> pH test.
Page 2 <strong>of</strong> 3<br />
<strong>Nitrazine</strong> pH analysis must be ordered by the patient’s physician.<br />
PATIENT IDENTIFICATION:<br />
Verify patient identification by checking the patient's name and date <strong>of</strong> birth on the arm band.<br />
PATIENT PREPARATION:<br />
Explain the purpose <strong>of</strong> the test and the steps <strong>of</strong> the procedure to the patient prior to performing<br />
the test.<br />
The operator’s hands must be washed before and after testing.<br />
Because <strong>of</strong> the hazardous nature <strong>of</strong> handling specimens contaminated with blood and body<br />
fluids, it is required that disposable latex gloves or an acceptable substitute be used when<br />
collecting specimens and test procedures. Standard precautions shall be observed for all<br />
specimens contaminated with blood and body fluid and they should be handled at Biosafety<br />
Level 2 as recommended for any potentially infectious material in the Centers for Disease<br />
Control/National Institutes <strong>of</strong> Health manual, Biosafety in Microbiological and Biomedical<br />
Laboratories, 1988 or in the National Committee for Clinical Laboratory Standard Document<br />
M29, Protection <strong>of</strong> Laboratory Workers from Infectious Disease Transmitted by Blood, Body<br />
Fluids, and Tissue, 1991.<br />
SUPPLIES: Gloves<br />
<strong>Nitrazine</strong> paper<br />
<strong>Nitrazine</strong> pH Quality Control buffers<br />
STORAGE:<br />
It is recommended that the <strong>Nitrazine</strong> pH paper be stored securely in its holder in a dry place at room<br />
temperature. <strong>Nitrazine</strong> pH papers expire on the date printed on the foil wrapper.<br />
QUALITY CONTROL:<br />
1. <strong>Nitrazine</strong> pH paper test strips should be tested when opening a new container and at least<br />
weekly using <strong>Nitrazine</strong> pH buffer controls, 5.0 and 7.0.<br />
2. Check the expiration date <strong>of</strong> the <strong>Nitrazine</strong> pH paper.<br />
3. Check the pH <strong>of</strong> each buffer; 5.0 and 7.0. Record the results on the <strong>Nitrazine</strong> pH QC Log.<br />
4. Note any failures on the <strong>Nitrazine</strong> pH QC Log. Retest; if failure again, obtain a new box <strong>of</strong><br />
<strong>Nitrazine</strong> pH paper and /or buffer(s) and follow steps= 1-4. Document corrective action<br />
taken on the QC Log.<br />
PROCEDURE:<br />
1. Explain procedure to woman/couple.<br />
2. Ensure that the fluid is appropriate for <strong>Nitrazine</strong> pH testing. Do not use for urine or gastric<br />
pH.<br />
3. Check the expiration date <strong>of</strong> the <strong>Nitrazine</strong> pH paper.<br />
4. Remove 3-4 inches <strong>of</strong> <strong>Nitrazine</strong> pH paper from container. With gloved hand, insert<br />
<strong>Nitrazine</strong> pH paper into the introitus <strong>of</strong> the vagina. Remove hand and paper.
Page 3 <strong>of</strong> 3<br />
INTERPRETATION OF COLOR CHANGE:<br />
COLOR pH INTERPRETATION<br />
yellow 5.0 membranes are probably not ruptured<br />
olive 5.5 “<br />
olive-green 6.0 “<br />
blue-green 6.5 membranes are probably ruptured<br />
blue-grey 7.0 “<br />
deep blue 7.5 may be due to blood or cervical mucous\<br />
**The <strong>Nitrazine</strong> pH values <strong>of</strong> blood, vaginal mucus, and certain secretions from vaginal infections are<br />
also alkaline. If the amount <strong>of</strong> amniotic fluid is small or absent but the above substances are present<br />
in large amounts, a false positive test could result. The <strong>Nitrazine</strong> pH test is not considered a definitive<br />
test for diagnosing ruptured membranes. Refer to the Fern Test for this.<br />
REPORTING RESULTS:<br />
1. Record patient results as positive or negative in the narrative nursing notes.<br />
2. Notify physician <strong>of</strong> results.<br />
3. The RN in charge <strong>of</strong> the patient will review , correlate, and assess results over<br />
each 24 hour period. The patient’s physician reviews results at a minimum each 24<br />
hours.<br />
COMPETENCY/CREDENTIALING<br />
The <strong>Nitrazine</strong> pH test competency certification/evaluation will be conducted annually. The<br />
Nurse Manager conducts the orientation training. The unit Nurse Manager or designee is<br />
responsible for overseeing this process <strong>of</strong> orientation and annual training. The training records<br />
are kept in the employee continuing education file. The nurse manager will submit a list <strong>of</strong><br />
certified operators and their dates <strong>of</strong> competency training each year to the Laboratory <strong>Point</strong> <strong>of</strong><br />
<strong>Care</strong> Coordinator.<br />
QUALITY ASSURANCE<br />
The Laboratory <strong>Point</strong> <strong>of</strong> <strong>Care</strong> Coordinator will submit a Quality Improvement Report to the<br />
Laboratory Director for PPM and Waived testing (nursing), Nurse Mangers, and DRH<br />
Laboratory Director quarterly.<br />
REFERENCE:<br />
Martin, Jean E, RN,(edited) Intrapartum, Management Modules- A Perinatal Education Program.<br />
<strong>Nitrazine</strong> <strong>Paper</strong> Test for Rupture Membranes. 1990.<br />
Procedure reviewed/approved by:<br />
___________________________________<br />
Victoria Orto, MS, RN, NEA-BC Date<br />
Chief Nursing Officer